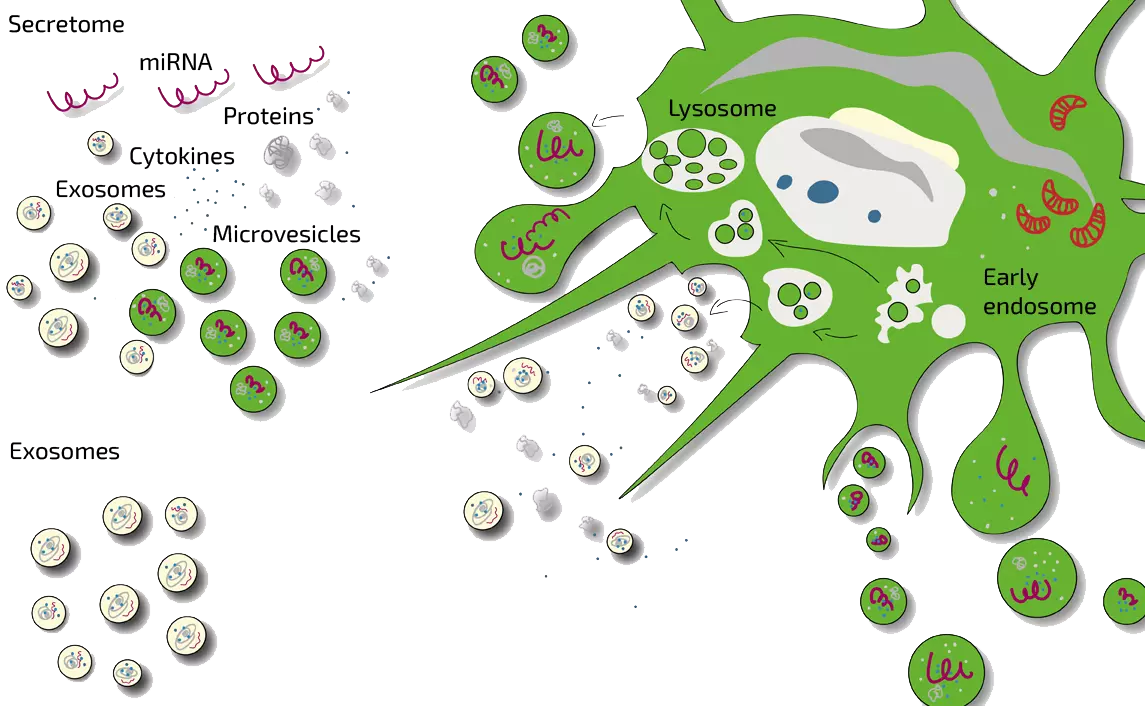
What is a Stem Cell Secretome? What are Exosomes ?
In scientific terms, the secretome is the totality of all substances released by stem cells to the outside. These include micro-vesicles, exosomes, proteins, cytokines (cell messengers), hormone-like substances and so-called immunomodulatory substances.
Based on current science, it is believed that the effect of stem cells in clinical trials conducted to date is not from the cells as such, but via the vesicles, protein molecules and other substances such as cytokines released by the cells. These substances released to the outside are called "the secretome" as an umbrella term, because the release is called secretion. Which substances of the secretome trigger the actual effects is still largely unknown. The ANOVA IRM secretome is produced from autologous mesenchymal stem cells.
.
Jump Directly to the Following Topics:
- Comparision of secretome and exosomes
- Advantages of a secretome therapy
- Work-flow of a secretome treatment
- Frequently asked questions
- Sources and literature
We Treat the Following Conditions with Secretome:
- Chronic inflammatory diseases
- Neurological disorders (MS, ALS, Parkinson)
- Poly-osteo-arthritis
- Rheumatoid arthritis
- Chronic inflammatory diseases
- Spinal cord transections (partial transections and, in combination with other therapies, complete transections)
- Erectile Dysfunktion, Impotence
- Anti-aging
Is Secretome the Same as Exosomes?
Secretome and exosomes are sometimes used as synonyms, but they are actually different.
The secretome is the totality of all messenger substances released to the outside, including the micro-vesicles and exosomes, but also other important factors that are dissolved in the fluid. These include many regeneration-promoting growth factors and so-called cytokines (cell-cell messengers).
Exosomes, on the other hand, are actually only the particulate exosomes that are isolated from the whole. Isolation concentrates them, but the important solutes and micro-vesicles are lost.
What is a Stem Cell Secretome Therapy?
ANOVA's stem cell secretome therapy is an experimental therapy that we use as part of individualized treatment for patients who may have a clear benefit from the therapy. In stem cell secretome therapy, stem cells are obtained from the patient's own adipose tissue (autologous stem cells, adipose stem cells, adipogenic stem cells, ADSCs). This initial small number of stem cells is grown and expanded (multiplied) in the pharmaceutical clean-room laboratory. This allows to obtain significantly more stem cells than e.g. in BMC without propagation. The goal of this process is to increase the number of stem cells, induce the production of the potentially regenerative and anti-inflammatory substances that stem cells release to the outside, concentrate them and use them for treatment. Instead of implanting or injecting stem cells into the patient, only the produced cellular components, i.e. the secretome, are applied. Compared to direct injection of stem cells, the risks of this method are relatively lower.
What are the Advantages of Treatment With
Stem Cell Secretome Compared to Stem Cells
In short, compared to BMC, pure mesenchymal stem cells are obtained and several doses can be obtained from one tissue collection, which makes it particularly suitable for the treatment of chronic diseases.
Moreover, the secretome is storable, fully quality controlled before use and is made from 90-98% pure MSC mesenchymal stem cells.
The use of stem cells is still limited in most countries. This has severely limited their use in medicine despite their presumed and in some cases clinically proven efficacy in treating many chronic or incurable diseases. Reasons for this include the fact that stem cells can lose their efficacy if they expand too much, altered cells can develop, and stem cells, at least those derived from blood (hematopoietic stem cells), are altered when frozen, which in turn alters their potential efficacy, including the composition of the substances they deliver to the outside world.
This may now be changing, with one of the latest breakthroughs in stem cell research: the ANOVA science team has developed and implemented a process that extracts and concentrates the components of stem cells that are evidence-based to be therapeutically active - the stem cell secretome.
The secretome that ANOVA uses in Offenbach provides safety in several respects.
No living cells are injected into the patient, i.e. no altered stem cells can be injected.
The secretome is made of mildly expanded (i.e. potentially less altered cells). Thus, the composition is the natural one, which is not altered by freezing.
After production, the secretome can be stored in this composition. Exosomes and other substances contained in the secretome remain stable. This allows to produce numerous doses with one withdrawal, which can then be used for more than 2 years.
Thus, treatment with secretome is the better option especially for chronic diseases.
.
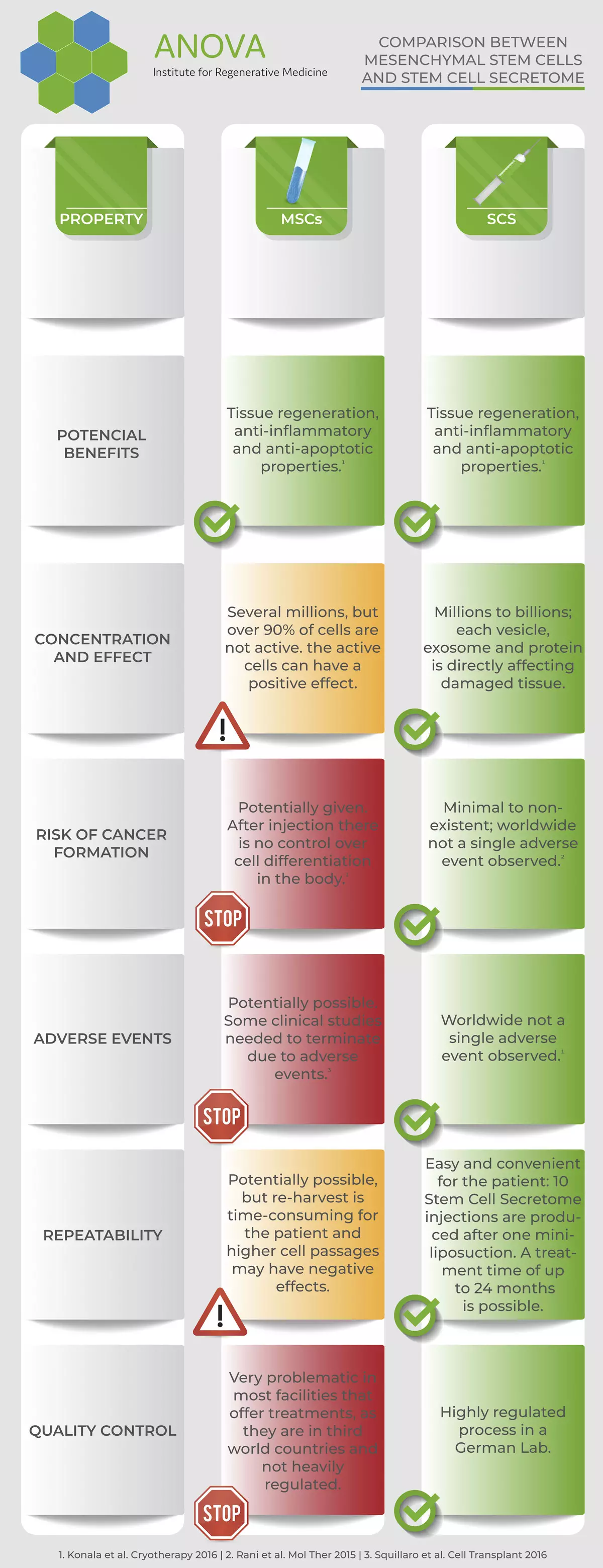
MSC and MSC secretome in comparison
Stem Cell Secretome Production and Application
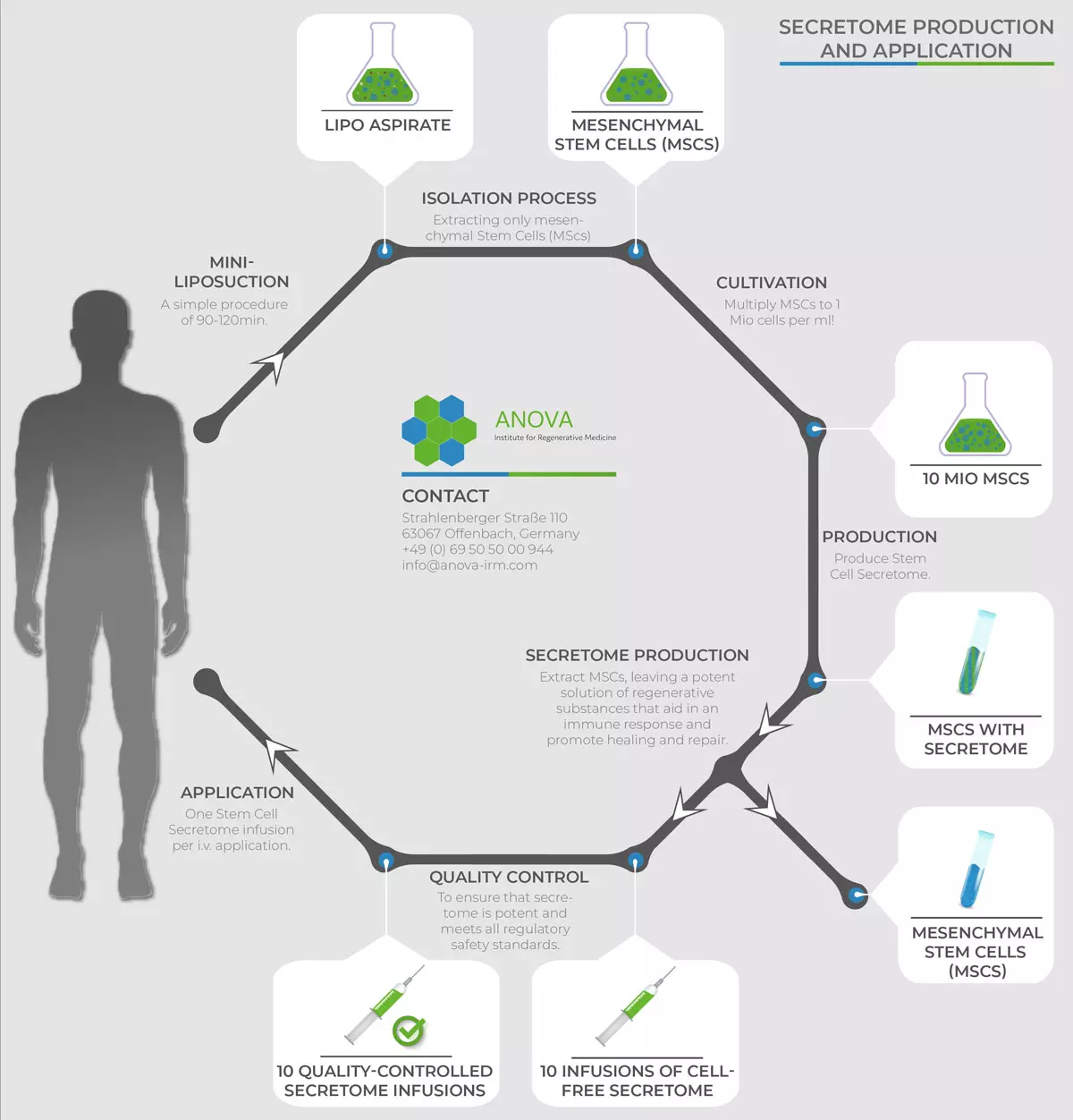
Secretome Production and Application
ANOVA IRM - Germany
Workflow of the Tissue Donation for Generation of MSC Secretome
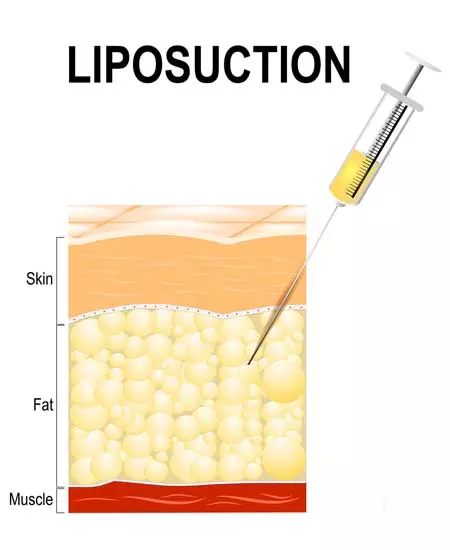
Liposuction
Step 1 - Harvesting Mesenchymal Stem Cells (MSCs) from subcutaneous adipose tissue
At ANOVA we use the so-called "nutational infrasonic liposuction", employing the Euromi Lipomatic® system. This procedure provides 10 to 100 times more viable MSCs obtained from the harvested adipose tissue than any other technique.
The adipose tissue is immediately processed in an ISO and GMP-certified clean-room guaranteeing the highest possible quality and safety of the end product.
Step 2 - Processing the stem cells and production of the Stem Cell Secretome (SCS)
In our specialized clean-room laboratories, the “stromal vascular fraction” (SVF) is isolated from adipose tissue after enzymatic digestion. The SVF is a mixture of different types of cells. It contains a high percentage of MSCs (equivalent of up to 500.000 cells per gram), and is one of the highest amounts of MSCs to be found in the human body.
In the next step, the cells are multiplied until they reach the requested quantity. By subjecting the cells to external stimuli, they are stimulated to secrete a high concentration of cytokines, micro-RNAs and growth factors (that retain regenerative abilities) that are packed in extracellular vesicles (micro-vesicles and exosomes). Together, these factors make up the secretome.
After a comprehensive quality control, the stem cell secretome is then ready for use, and can be stored for several months at minus 80°C. This makes it possible to design a therapy plan that allows for several infusions (over 2 years) that favors optimal treatment outcomes.

Production

Application
Step 3 - Application and treatment with ANOVA's Stem Cell Secretome therapy
From a single isolation of stem cells via liposuction of the patient's adipose tissue (step 1), up to 10 therapeutic doses of Stem Cell Secretome can be obtained that are to be administered systemically via infusion.
The earliest application of Stem Cell Secretome is possible 4 weeks after liposuction. During that time, our specialists in the laboratory prepare the product and perform quality controls.
Individual Treatment Plans - Injections
When creating your treatment plan, both the disease-specific factors and your specific situation as a patient are taken into account. In general, 10 doses are available after liposuction. Depending on the disease and stage, the treatments for acute diseases tend to take place in quick succession (intense, figure below), for chronic diseases at regular intervals. For patients from Germany and surrounding countries such as Austria, Switzerland, Italy, the Netherlands, Great Britain, France, Spain, etc., it is usually easy and inexpensive to travel several times. Therefore, injections are usually scheduled once as multiple doses and subsequently as single injections 6-8 weeks apart (boost+balanced). In contrast, for patients from distant regions such as the USA, Canada, South America, Brazil, the Middle East, Saudi Arabia, Oman, Russia, etc., treatments are usually scheduled continuously as multiple doses but at longer intervals to reduce the number of trips.
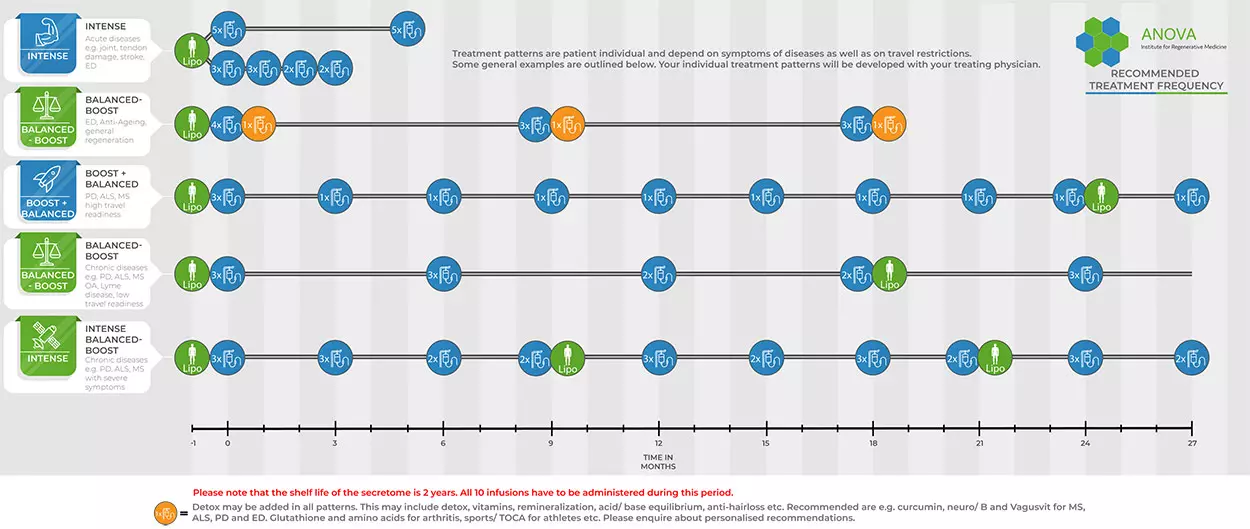
Secretome Treatment Plan ANOVA IRM Germany
Treatment Workflow in Detail
The scheme below summarizes all steps towards your individual therapy at ANOVA IRM. It starts from your first charge-free contacts and inquiries to ANOVA including the initial analysis whether our therapies are suitable for treatment of your condition and if you are eligible as a donor. Continues with financial issues such as cost-estimate and treatment contracts, virtual or in person doctoral appointments, your trip to Offenbach, Germany, testing, physical exams, tissue donation, stem cell product cultivation and quality control up to application.
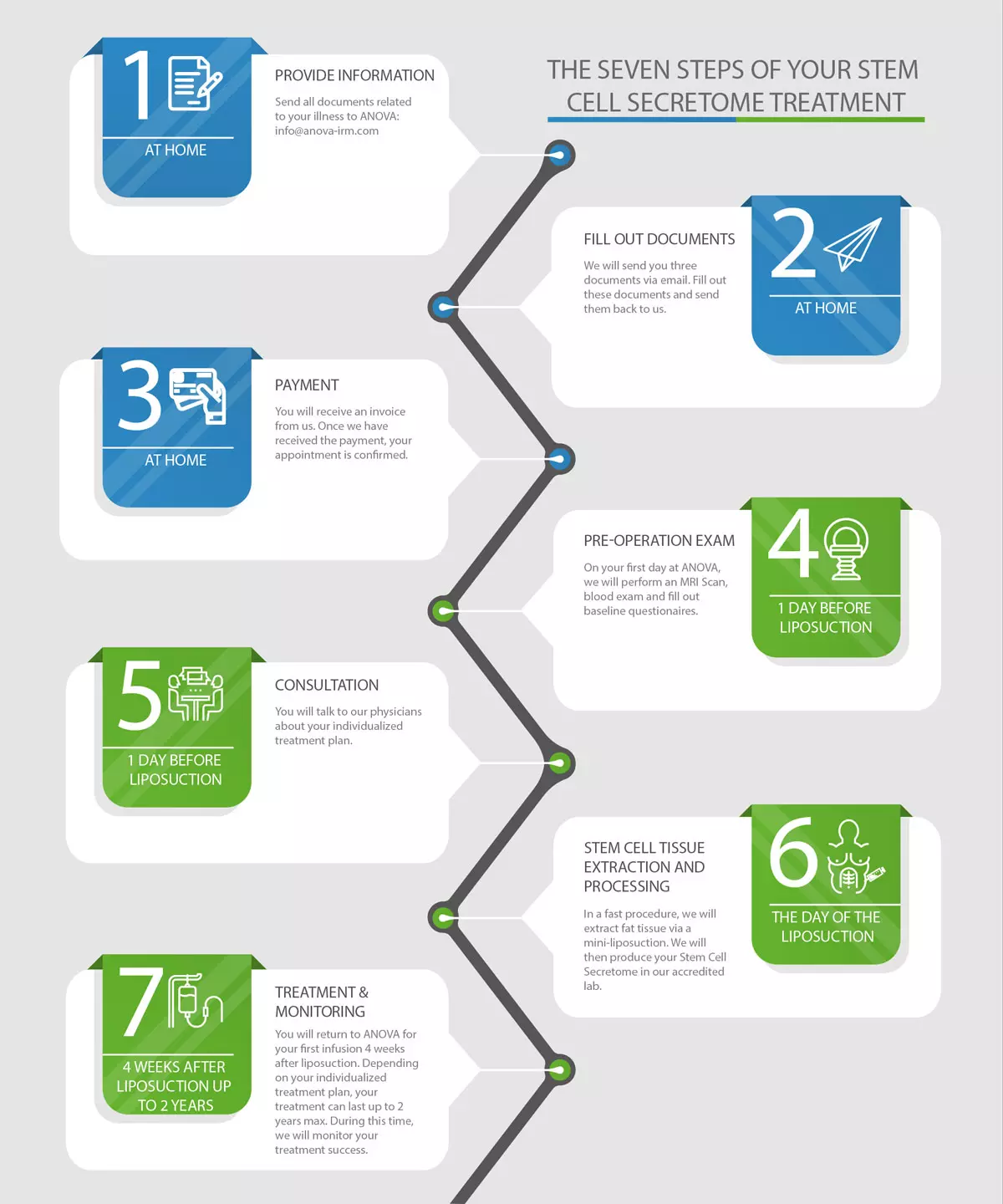
The 7 Steps of your Stem Cell Therapy
Contraindications
Our stem cell treatments are experimental, but we only treat patients for whom we believe the risk/benefit ratio indicates treatment based on the state of the art, i.e., medical, scientific evidence.
Please understand that we therefore do not treat patients for whom the following points apply:
- Active cancer in the last two years
- Not yet of legal age
- Existing pregnancy or lactation period
- Unable to breathe on own, ventilator
- Difficulty breathing in supine position
- Dysphagia (extreme difficulty swallowing)
- Psychiatric disorder
- Active infectious disease (Hepatitis A, B, C, HIV, Syphilis, or other)
ANOVA Stem Cell Secretome Therapy: Advantages and Applications
Advantages
- The Secretome is devoid of all cells, which will decrease the risk of unwanted reactions in allogenic use.
- The Secretome has been tested in an autologous setting in hundreds of patients without any detrimental side effects.
- Therapeutic doses can be achieved with as little as 1 million of mesenchymal stem cells.
- The Stem Cell Secretome, once produced, is ready for use and can be stored at -80°C for up to 2 years.
- Up to ten therapeutic doses of Stem Cell Secretome are available for a therapy course without renewed stem cell isolation.
- ANOVA Stem Cell Secretome therapy can be carried out at regular intervals over extended periods of time, e.g. at weekly intervals, providing a scheduled treatment plan instead of a single treatment at only one time point.
Applications
- ANOVA's Stem Cell Secretome therapy is a novel therapy with regenerative effects in:
- Autoimmune Diseases:
- Neurodegenerative Diseases:
- Multiple Sclerosis (MS)
- Parkinson’s Disease (PD)
- Alzheimer’s Disease (AD)
- Amyotrophic Lateral Sclerosis (ALS)
- Orthopedics and Sport Injuries:
- Erectile Dysfunction, Impotence and Incontinence
- Spinal Cord Injury (SCI)
- Stroke
- Cardiovascular Diseases
- Liver and Kidney Diseases
- and a few more upon request and after thorough evaluation
FAQ - Frequently Asked Questions - Exosomes - Secretome
More Information on exosomes is also given in your blog post: The Power of Exosomes.
Vocabulary and Definitions
Differentiation - Stem cells have the potential to differentiate into different cell types depending on external stimuli and signals. MSC e.g. must be able to become at least bone cells, cartilage cells and fat cells to be called stem cell.
Cancer, cancer cell - cells in which the normal regulation of cell division is no longer functional. These cells divide even though this would not be necessary and thus continue to multiply to the detriment of the organism.
Morphology - The shape, appearance of cells under the microscope.
Proliferation - cell division, cell multiplication
Stem cell - a cell that has a greater potential in its reproduction than normal body cells. It can renew itself and differentiate into several cell types = functionalize.
What is a Secretome?
Stem cells communicate with other cells by releasing messenger substances to the outside. The totality of all released substances is called secretome (from secretion = release to the outside). The secretome can contain exosomes, micro-vesicles, proteins, growth factors, hormones, cytokines and other substances. A scientific review article on vesicles can be found here.
What is an Exosome?
Exosomes are small messenger packages that cells release to the outside.They ar a part of the secretome.
Exosomes are approximately 30-100nm (i.e. 0.03 to 0.1mm) in size and consist of an outer double-lipid layer (like liposomes) and an interior that can be loaded with fluid and messengers such as miRNA and proteins. Exosomes are released to the outside by the fusion of multi-vesicular corpuscles inside the cell with the cell membrane. There, they can be taken up by other cells to mediate reactions. A scientific review article on vesicles can be found here.
What are Micro-Vesicles?
Micro-vesicles are also lipid membrane-coated particles that can carry cargo. However, at 100 to 1,000nm in size, they are larger than exosomes and are released to the outside by pinching off (exocytosis) at the cell membrane. A scientific review article on vesicles can be found at hier.
What are Growth Factors?
Chemically, growth factors usually are proteins that bind to receptors on the outer surface of target cells. Many cells produce growth factors such as EGF (epidermal growth factor), PDGF (platelet-derived growth factor), NGF (neuronal growth factor), bFGF (basic fibroblast growth factor) etc. Growth factors in general are not related to Human growth hormone (HGH).
Cells respond to their environment. They divide only when necessary. In a healthy body, for example, a liver cell divides only once a year to replace dead cells. The signal for a cell to divide can be the loss of neighboring cells (cut) or growth factors produced by other cells. These factors diffuse to other cells, bind to receptors on the surface, and trigger signaling cascades inside the target cell that stimulate division. Inflammation often inhibits cell division (proliferation).
How is Secretome Produced? What Tissue is Used for Secretome Production?
The secretome used by the ANOVA Institute for Regenerative Medicine is produced from mesenchymal stem cells of the patient (autologous = own stem cells). These are extracted from the abdominal fat in a short, mini-liposuction and subsequently cultivated very gently and thus propagated.
After fat harvesting, the starting material is the SVF (stromal vascular fraction) isolated from it, which contains only about 1-10% MSC and progenitor cells. Subsequently, the MSC are cultivated, multiplied and a pure culture consisting of 90-98% MSC is obtained.
The fact that these cells are division-active stem cells has been proven by a so-called multi-differentiation assay. In this, the MSC are differentiated into cell types (cartilage cells, bone cells and fat cells) over 2-3 weeks. If this is successful, we have one of several proofs that the cells are indeed stem cells. In addition, we check each batch for purity.
What are the Advantages of Secretome?
The great advantage of the secretome is that it can be frozen. If stem cells are frozen, their properties change and this can lead to a loss of efficacy. In the case of chronic diseases, however, a continuous therapy is needed, i.e. stem cells would have to be taken again and again. In the case of secretome, one can obtain 10-20 doses from one collection and thus treat repeatedly over a period of 2 years.
Another advantage is that MSC (mesenchymal stem cells, stromal cells), which are cultivated over a longer period of time, bear the risk of cancer cells developing in the culture. If these cells are then used, they will be transferred to the patient. This risk does not exist with the ANOVA secretome, as it is cell-free and tested.
More Stem Cells - More Effect? Older Stem Cells - Lower Efficacy!
Many patients believe that a larger amount of stem cells also achieves better effects. This is correct up to a limit that is difficult to define. However, since it is not yet possible to measure "efficacy", or only in very few cases, one should be very cautious here. Many characteristics of stem cells are not well understood so far and nobody knows exactly which factors and substances define an effect. The only thing that is really known exactly is that with increasing multiplication (generations, population doublings) the efficacy potential decreases. That is, the more you multiply stem cells, the less likely they are to still be effective.
ANOVA therefore multiplies the cells only within very conservative limits internationally recognized by research and science, so that the risk of potential loss of efficacy is kept extremely low.
Stem Cell Therapies sorted by stem cell type (source tissue) and product
References and Literature - Stem Cell Secretome
- Konala, Vijay Bhaskar Reddy, et al. "The current landscape of the mesenchymal stromal cell secretome: a new paradigm for cell-free regeneration." Cytotherapy 18.1 (2016): 13-24.
- Lopez-Verrilli, M. A., et al. "Mesenchymal stem cell-derived exosomes from different sources selectively promote neuritic outgrowth." Neuroscience 320 (2016): 129-139.
- Kim, Hyun Ok, Seong-Mi Choi, and Han-Soo Kim. "Mesenchymal stem cell-derived secretome and microvesicles as a cell-free therapeutics for neurodegenerative disorders." Tissue Engineering and Regenerative Medicine 10.3 (2013): 93-101.
- Rani, Sweta, et al. "Mesenchymal stem cell-derived extracellular vesicles: toward cell-free therapeutic applications." Molecular Therapy 23.5 (2015): 812-823.
- Zhang, Xiaoyan, et al. "Mesenchymal Stem Cell-Derived Extracellular Vesicles: Roles in Tumor Growth, Progression, and Drug Resistance." Stem Cells International 2017 (2017).
- omzikova, Marina O., and Albert A. Rizvanov. "Current Trends in Regenerative Medicine: From Cell to Cell-Free Therapy." BioNanoScience (2016): 1-6.
- Zhang, Bin, et al. "Focus on extracellular vesicles: Therapeutic potential of stem cell-derived extracellular Vesicles." International journal of molecular sciences 17.2 (2016): 174.
- Katsuda T. et al. (2013). Human adipose tissue-derived mesenchymal stem cells secrete functional neprilysin-bound exosomes. Scientific reports, 3, 1197.
- Pusic A. D. et al. (2014). IFN?-stimulated dendritic cell exosomes as a potential therapeutic for remyelination. Journal of neuroimmunology, 266(1), 12-23.
- Maumus M, Jorgensen C, Noël D (2013) Mesenchymal stem cells in regenerative medicine applied to rheumatic diseases: Role of secretome and exosomes. Biochimie 95:2229?2234. doi: 10.1016/j.biochi.2013.04.017
- Drago D, Cossetti C, Iraci N, et al (2013) Biochimie The stem cell secretome and its role in brain repair. Biochimie 95:2271?2285. doi: 10.1016/j.biochi.2013.06.020
- Sevivas N, Teixeira FG, Portugal R, et al (2016) Mesenchymal Stem Cell Secretome: A Potential Tool for the Prevention of Muscle Degenerative Changes Associated With Chronic Rotator Cuff Tears. Am J Sports Med. doi: 10.1177/0363546516657827
- Hs K (2016) Mesenchymal Stem Cells vs . Mesenchymal Stem Cell Secretome for Rheumatoid Arthritis Treatment. 1:1?2.
- Maumus M, Jorgensen C, Noël D (2013) Mesenchymal stem cells in regenerative medicine applied to rheumatic diseases: Role of secretome and exosomes. Biochimie 95:2229?2234. doi: 10.1016/j.biochi.2013.04.017
- Kapur SK, Katz AJ (2013) Biochimie Review of the adipose derived stem cell secretome. Biochimie 95:2222?2228. doi: 10.1016/j.biochi.2013.06.001
- Ranganath SH, Levy O, Inamdar MS, Karp JM (2012) Harnessing the mesenchymal stem cell secretome for the treatment of cardiovascular disease. Cell Stem Cell 10:244?258. doi: 10.1016/j.stem.2012.02.005
- Tran C, Damaser MS (2015) Stem cells as drug delivery methods: Application of stem cell secretome for regeneration. Adv Drug Deliv Rev 82:1?11. doi: 10.1016/j.addr.2014.10.007
- Zimmerlin L, Park TS, Zambidis ET, et al (2013) Mesenchymal stem cell secretome and regenerative therapy after cancer. Biochimie 95:2235?2245. doi: 10.1016/j.biochi.2013.05.010
- Calamia V, Lourido L, Fernandez-Puente P, et al (2012) Secretome analysis of chondroitin sulfate-treated chondrocytes reveals its anti-angiogenic, anti-inflammatory and anti-catabolic properties. Arthritis Res Ther 14:R202. doi: 10.1186/ar4040
- Ranganath SH, Levy O, Inamdar MS, Karp JM (2012) Review Harnessing the Mesenchymal Stem Cell Secretome for the Treatment of Cardiovascular Disease. Stem Cell 10:244?258. doi: 10.1016/j.stem.2012.02.005
- Teixeira FG, Carvalho MM, Sousa N, Salgado AJ (2013) Mesenchymal stem cells secretome: A new paradigm for central nervous system regeneration? Cell Mol Life Sci 70:3871?3882. doi: 10.1007/s00018-013-1290-8
- Kapur SK, Katz AJ (2013) Review of the adipose derived stem cell secretome. Biochimie 95:2222?2228. doi: 10.1016/j.biochi.2013.06.001
- Chang C-P, Chio C-C, Cheong C-U, et al (2013) Hypoxic preconditioning enhances the therapeutic potential of the secretome from cultured human mesenchymal stem cells in experimental traumatic brain injury. Clin Sci (Lond) 124:165?76. doi: 10.1042/CS20120226
- Salgado AJ, Sousa JC, Costa BM, et al (2015) Mesenchymal stem cells secretome as a modulator of the neurogenic niche: basic insights and therapeutic opportunities. Front Cell Neurosci 9:1?18. doi: 10.3389/fncel.2015.00249
- Ahmed NE-MB, Murakami M, Hirose Y, Nakashima M (2016) Therapeutic Potential of Dental Pulp Stem Cell Secretome for Alzheimer?s Disease Treatment: An In Vitro Study. Stem Cells Int 2016:8102478. doi: 10.1155/2016/8102478
- Bhaskar V, Konala R, Mamidi MK, et al (2016) The current landscape of the mesenchymal stromal cell secretome : A new paradigm for cell-free regeneration. Cytotherapy 18:13?24. doi: 10.1016/j.jcyt.2015.10.008
- Malda J, Boere J, van de Lest C, et al (2016) Extracellular vesicles - new tool for joint repair and regeneration - IN PRESS. Nat Rev Rheumatol 12:243?249. doi: 10.1038/nrrheum.2015.170
- Lener T, Gimona M, Aigner L, et al (2015) Applying extracellular vesicles based therapeutics in clinical trials Á an ISEV position paper. 1:1?31.
- Dostert G, Mesure B, Menu P, Velot É (2017) How Do Mesenchymal Stem Cells Influence or Are Influenced by Microenvironment through Extracellular Vesicles Communication ? 5:1?7. doi: 10.3389/fcell.2017.00006
- Joshi P, Benussi L, Furlan R, et al (2015) Extracellular vesicles in Alzheimer?s disease: Friends or foes? focus on A?-vesicle interaction. Int. J. Mol. Sci. 16:4800?4813.
- Gao T, Guo W, Chen M, et al (2016) Extracellular Vesicles and Autophagy in Osteoarthritis.
- Katsuda T, Ochiya T (2015) Molecular signatures of mesenchymal stem cell-derived extracellular vesicle-mediated tissue repair. Stem Cell Res Ther 6:212. doi: 10.1186/s13287-015-0214-y
- Lener T, Gioma M, Aigner L, et al (2015) Applying extracellular vesicles based therapeutics in clinical trials - an ISEV position paper. J Extracell Vesicles 4:1?31. doi: 10.3402/jev.v4.30087
- Xu Y, Guo S, Wei C, et al (2016) The Comparison of Adipose Stem Cell and Placental Stem Cell in Secretion Characteristics and in Facial Antiaging.
- Buul GM Van, Villafuertes E, Bos PK, et al (2012) Mesenchymal stem cells secrete factors that inhibit in fl ammatory processes in short-term osteoarthritic synovium and cartilage explant culture. Osteoarthr Cartil 20:1186?1196. doi: 10.1016/j.joca.2012.06.003
- Baglio SR, Pegtel DM, Baldini N (2012) Mesenchymal stem cell secreted vesicles provide novel opportunities in ( stem ) cell-free therapy. 3:1?11. doi: 10.3389/fphys.2012.00359
- Anderson JD, Pham MT, Contreras Z, et al (2016) Mesenchymal stem cell-based therapy for ischemic stroke. Chinese Neurosurg J 2:36. doi: 10.1186/s41016-016-0053-4
- Biology C, Cell R, Eye N, Institutes N (2017) Bone Marrow-Derived Mesenchymal Stem Cells-Derived Exosomes Promote Survival of Retinal Ganglion Cells Through miRNA-Dependent Mechanisms. 1273?1285.