Promising New Therapy for Multiple Sclerosis with Stem Cell Secretome
at ANOVA IRM in Offenbach, Germany
Multiple Sclerosis is caused by a misguided autoimmune response (attack) against the “myelin sheath” of nerve cells. The myelin sheath is much like an insulation layer of a cable. Its function is to electrically insulate the nerve, to protect it, and to ensure the proper function of the nervous system (see Fig. 1). Damage or degeneration of this protective layer will lead to impairments to the normal function of the affected nerves. The severity of impairments varies among individuals.
ANOVA utilizes the Stem Cell Secretome, a therapeutic which is made of paracrine factors of stem cells with the goal to achieve the above mentioned effects. In Europe, we are the first clinic to offer this treatment.
According to the regenerative effects of Mesenchymal Stem Cells on the nervous and immune system, all types of Multiple Sclerosis can be treated effectively, such as: Relapsing Remitting MS (RRMS), Primary Progressive MS (PPMS) and Secondary Progressive MS (SPMS).
To find out more about ANOVA's Stem Cell-based Multiple Sclerosis treatment plans and guidelines, schedule an appointment today.
Multiple Sclerosis
Diagnostics - Treatment - Medication - Stem Cell Therapies
On this page we inform you about MS covering an overview on important aspects of causes, treatment options, precision diagnostics as well as our stem cell-based therapies that we offer in Offenbach (near Frankfurt am Main airport), Germany.
Jump directly to the following topics:
- Conventional therapies
- ANOVA therapies for MS
- Expectations and limits
- Our treatment options
- Workflow of the treatment process
- FAQ- frequently-asked questions
- Sources and Literature
Conventional MS Therapies
Current drugs and medications for Multiple Sclerosis (MS) are administered to slow down the progression of the disease and reduce its symptoms. These drugs are known to suppress the general immune responses that lead to neurological symptoms of MS. The reversal of previous symptoms and preventing the disease's progression is rarely achieved. Therapy options like physiotherapy and muscle relaxants (such as the alternative canabinoid treatment) help the patients immensely but also do not slow the progression or restore function.
Besides, the immuno-supressants used have multiple side-effects and in many cases interferon-treatments shift the relapsing-remitting pattern to a secondary progressive pattern which has severe effects on the quality of life. Many patients stop interferon treatment due to this effect.
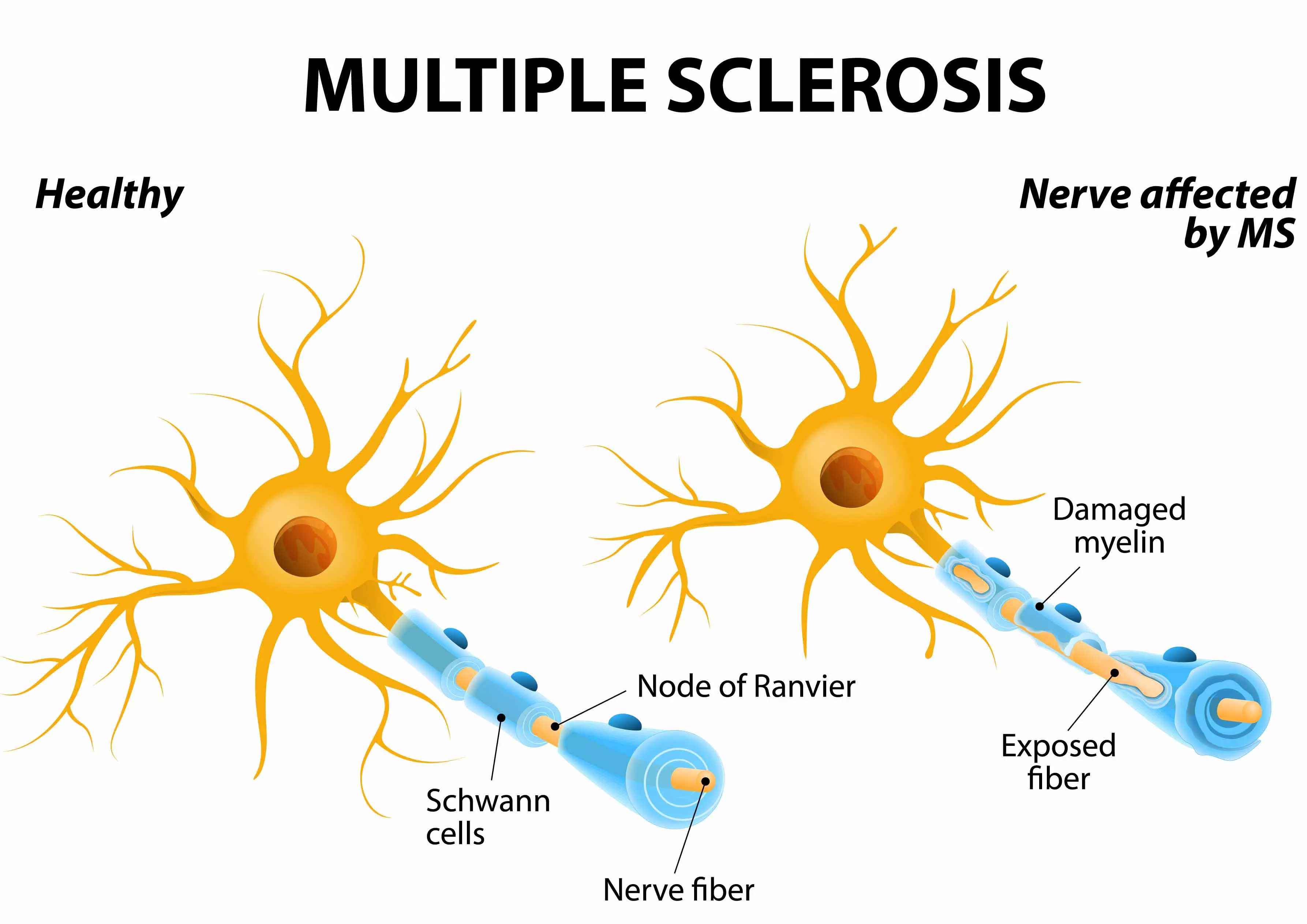
Myelin damage in Multiple Sclerosis
ANOVA IRM - Germany
Stem Cell Therapies for MS
As such, stem cell-based therapies have recently emerged as a promising novel treatment option for MS. Stem cells have shown to have beneficial effects on the immune system, but also stimulate regeneration of the neurological damages that are induced by MS. Therefore, stem cell-based therapies are an emerging option to treat the disease. Their natural capacity to modulate the immune system is based on cell-cell communication by soluble factors and this natural type of regulation might prove more beneficial in the future even though stem cell too, are no cure for MS.
Stem cells appear to have two major beneficial effects for treating this severe autoimmune, neurodegenerative disease, as they were shown to:
- Modify the effects of the immune system, such as protection from further attacks on the myelin sheaths (neuroprotection)
- Repair damaged nerves (remyelination)
Treating MS patients with stem cell-based therapies has already been conducted with much success in external clinical trials. The promising results from these phase I/II studies have shown to:
- Reduce inflammation
- Improve visual acuity and evoke improved response latency
- Increase of optic nerve area
- Inhibition of new lesion development and reduction of lesion areas
- Reduce incidences of relapse as compared to patients who received anti-inflammatory and immuno-suppression treatments only
Most importantly, until now, no major side effects have been noted in treating MS patients with stem cells.
Stem cell research has allowed ANOVA, a German Stem Cell Clinic in the heart of Europe near Frankfurt/Main airport, to offer a novel treatment with a new therapeutic approach: The ANOVA Stem Cell Secretome is a cell free and promising treatment option for MS.
Check your eligibility, apply for a treatment or simply write to receive more information.
Stem Cell Treatments for Multiple Sclerosis at
ANOVA Institute for Regenerative Medicine - Offenbach, Germany
Secretome/Exosomes of MSC
Potency Hypothesis of Stem Cell Therapies
Stem cells possess the potential to communicate with the immune cells that elicit inflammation and by natural, so far not understood mechanisms inhibit immune-over-reactions. Furthermore, stem cells have the ability to stimulate regeneration of tissue thereby counteracting the loss of function.
MSEC - Mesenchymal Stem Cell Secretome - Exosomes - Autologous
As MS is a chronic, so far not curable disease, we on-goingly treat patients with MS in early to mid stages with MSEC (secretome, exosomes, EVs) of mesenchymal stem cells (MSC, AD-MSC, adipose-derived, fat-derived stem cells) which we harvest from the patients belly in a mini-liposuction (very brief and limited liposuction) under slight sedation. Worldwide, ANOVA is the first stem cell clinic to acquire legal permission form the responsible governmental authorities and therefore, offers high quality, safe and legally-controlled autologous (own) exosome-containing secretome.
The main advantage of MSEC is that in contrast to live stem cells which would loose their therapeutic potency, can be frozen without loss of exosomes. This enables us to produce 10-20 injection doses from one liposuction which can then be administered over a longer treatment period. This is especially advantageous for repeated immuno-modulation, stimulation of cell survival and regeneration in MS. What a Secretome/Exosome is and how they compare is explained on our overview page.
Please note that this treatment is not a cure but as any stem cell treatment an experimental, potentially disease-modifying therapy. It requires regular and repeated travelling to Offenbach, Germany.
Contraindications
Our stem cell treatments are experimental, but we only treat patients for whom we believe the risk/benefit ratio indicates treatment based on the state of the art, i.e., medical, scientific evidence.
Please understand that we therefore do not treat patients for whom the following points apply:
- Active cancer in the last two years
- Not yet of legal age
- Existing pregnancy or lactation period
- Unable to breathe on own, ventilator
- Difficulty breathing in supine position
- Dysphagia (extreme difficulty swallowing)
- Psychiatric disorder
- Active infectious disease (Hepatitis A, B, C, HIV, Syphilis, or other)
Therapy Workflow for Early to mid-Stage Multiple Sclerosis
The precise workflow is described in detail on the stem cell- specific page of Secretome/Exosomes.
All therapies are divided into phases such as evaluation of the medical history (we analyze your current therapies and medical records), initial counseling and evaluation of potential, patient-individual benefit of a stem cell therapy (indication statement), preliminary examinations, diagnostics, consultation on all therapy options, preparation of an individual treatment plan including cost estimate, harvesting of tissue, production of the stem cell product, quality control of the product and application.
Unfortunately, we only treat patients in an early to mid-stage of MS. Patients need to be able to breathe unassitedly and need to be fit for sedation and the brief intervention needed to collect fat. According to the risk-benefit ratio, we cannot treat children or pregnant women. In addition, other factors can also be exclusion criteria.
How Long Does a Stem Cell Therapy Take?
The initial analyses and counseling can be done without you having to travel to Offenbach (near Frankfurt/Main, Germany). This period can be 2 weeks up to months depending on the availability of patients slots. If you live further away, we will conduct the initial discussions by telephone or video conference. For the actual treatment, you will travel to Offenbach.
Secretome/Exosome-therapy:
Preparation and harvest of the fat (mini-liposuction) need once 2 days (consecutive days) in Offenbach, followed by enrichment of the mesenchymal stem cells (Secretome/Exosome) and quality control. Approximately 4 weeks after the isolation, the therapy begins according to the therapy plan determined with you. You will then come to Offenbach am Main (Germany) in regular intervals for the application. Depending on where you live and your traveling capacity and restrictions the treatment pattern is adjusted to your needs and abilities. The shelf life of the secretome (exosomes) is 2 years.
As MS is not curable with any treatment, we recommend a double-lipo which produces 20 doses for a continuous treatment over 2 years. Thereafter, a new liposuction has to be performed.
How Much Does Stem Cell Treatment Cost?
Our treatments are always tailored to your specific situation, disease, stage and other factors. The therapies differ in the product used (BMC, secretome, PRP or hyaluronic acid), the frequency of treatment as well as the further examinations and your sedation and anesthesia wishes. A treatment for MS will cost well above ten thousand euros. You will receive a cost estimate for all treatments in advance so that you can accurately estimate what a treatment would cost in your individual case.
Does my Health Insurance Cover the Therapy Costs?
Unfortunately, at the moment it is assumed that health insurance companies do not cover the costs of experimental therapies (BMC, secretome, PRP, micro-fracture technique), i.e. you will have to bear the costs entirely yourself.
Treating Early-to-Mid-Stage Multiple Sclerosis at ANOVA
Our team focuses on offering our patients a personalized treatment in regenerative medicine. We aim to combine well-established therapies with novel and promising therapies to obtain the best possible outcome. Just as importantly, a full diagnostic work-up is a necessity to fully understand the patient’s current state. The combination with diagnostics with state-of-the-art technology and evidence-based effective therapies make our treatments so special. But, as with any type of treatment, experimental therapy such as stem cell-based therapy cannot promise any success. Before the attending physician can suggest an experimental therapy, he or she must individually check whether benefits of the treatment are given for the patient and whether these benefits outweigh the potential risks. Make an appointment today to learn more about your treatment options at ANOVA.
Stem Cell-based Treatment for Multiple Sclerosis:
Your Personalized High Quality Treatment
ANOVA uses the knowledge of recent scientific research to produce and combine effective stem cell therapies.ANOVA IRM has legal permits for all its therapeutics and is bi-annually controlled by the German authorities. All products undergo pharmaceutical grade quality controls that go beyond legal requirements.
Furthermore, we have developed and applied standardized procedures for stem cell isolation and processing, as well as diagnostic work-up. This includes, amongst others, the use Magnetic Resonance Imaging (MRI), coupled with extensive blood examination. In addition to our advanced diagnostics, we objectively track the therapeutic success of the stem cell-based treatment after its administration. We aim to provide the best personalized stem cell-based therapy for patients that seek treatment for MS.
Stem cell therapy is an experimental therapy. As such, it is important that your doctor assesses the benefits and the risks for your individual case. If the benefits outweigh the risks, your doctor can suggest stem cell therapy as an option to you. To find out more about your treatment options at ANOVA Institute for Regenerative Medicine, a private German clinic, please click here to contact us.
FAQ: Stem Cell-based Therapies for Multiple Sclerosis
What is Multiple Sclerosis?
Multiple sclerosis (MS) is a disease of the central nervous system, which includes the spinal cord and the brain. In patients with MS, the immune system attacks the protective sheath which is wrapped around nerve fibers. This sheath is called myelin. This autoimmune attack causes nervous system damage and can lead to communication issues between the brain and rest of your body.
MS is potentially disabling because it can cause permanent damage to the nerves.
There is currently no treatment which can cure MS. However, there are some therapies which can potentially help improve recovery times after attacks, alter disease course and manage the symptoms.
What are the Symptoms of Multiple Sclerosis?
Multiple sclerosis is unfortunately one of those diseases which has a variety of symptoms, making it difficult for potential patients to immediately identify. They also differ greatly from patient to patient, and can change with time. All of this is due to the fact that different nerve fibers can be affected, and with that, the symptoms can be different.
A lot of symptoms relate to movement impairment; examples are:
- Numbness / weakness in the limbs. This can also occur on one side of the body at a time
- Tremor and lack of coordination, including unsteady gait
- Uncomfortable electrical sensations which happen with certain neck movements, especially bending the neck forward (Lhermitte sign)
- Tingling / pain in different parts of the body
Some symptoms relate to vision impairment; examples are:
- Loss of vision, often in one eye at a time
- Pain during eye movement
- Double vision for an extended amount of time
- Blurry vision
Other symptoms can include:
- Fatigue and dizziness
- Impairment of speech
- Impairments in sexual, continence and bowel functions
What causes Multiple Sclerosis and What are the Risk Factors?
MS is considered an autoimmune disease, because the patient’s own immune system attacks part of the body, in this case the nerve fibers. Unfortunately, the causes of MS are unknown.
The current scientific hypothesis is that the underlying mechanism is either related to destruction by the immune system or failure of the myelin-producing cells.
Scientists believe that it may be a combination of environmental factors, such as a viral infection, and genetics which is responsible for the disease.
Factors that may increase the risk of developing MS:
- Age: While the disease can develop in patients of any age, it often develops between the ages of 20 and 40 years.
- Sex: Women are 2 to 3x as likely to develop relapsing-remitting MS compared to men.
- Family history: The risk to develop MS is higher if parents or siblings have MS.
- Specific viral infections: Some viruses have been linked to MS, such as Epstein-Barr.
- Race: White people are at highest risk of developing MS, which people of Asian, African or Native American descent have the lowest risk.
- Vitamin D deficiency: The risk to develop MS is higher when there are low levels of vitamin D and/or low exposure to sunlight.
Types of Multiple Sclerosis, Disease Course and Complications
Several types and patterns of progression have been described. The classification of the disease takes into account the past course to predict the future course. This is necessary to be able to determine prognosis and to make sound treatment decisions.
Currently, four types of MS can be described:
- Clinically isolated syndrome (CIS)
- Relapsing-remitting MS (RRMS)
- Primary progressive MS (PPMS)
- Secondary progressive MS (SPMS)
Most patients with MS initially experience the relapsing-remitting type, which is characterized by unpredictable relapses followed by longer time periods of remission, when symptoms partially or completely improve. During remission, there are no new signs of disease activity. The relapsing-remitting type of MS usually begins with a clinically isolated syndrome (CIS), where a person has an attack suggestive of demyelination, but does not fulfill the criteria for MS.
In 10 to 20% of patients, primary progressive MS occurs. Here, there is no remission after the initial symptoms and is characterized by progression of disability from onset, with no or minor remissions and improvements. Secondary progressive MS occurs in at least half of those with initial relapsing-remitting disease. When this happens, patients will develop progressive neurologic decline between acute attacks without any definite periods of remission. In these cases, occasional relapses and minor remissions may appear. Disease progression is very different from patient to patient when they deal with secondary-progressive MS.
MS can unfortunately cause a lot of other complications as well. Multiple sclerosis patients may develop:
- Paralysis
- Epilepsy
- Muscle stiffness / spasms
- Forgetfulness, mood swings, depression and other mental impairments
Why Modern Medicine Should use Stem Cells for Multiple Sclerosis -
The Scientific Background:
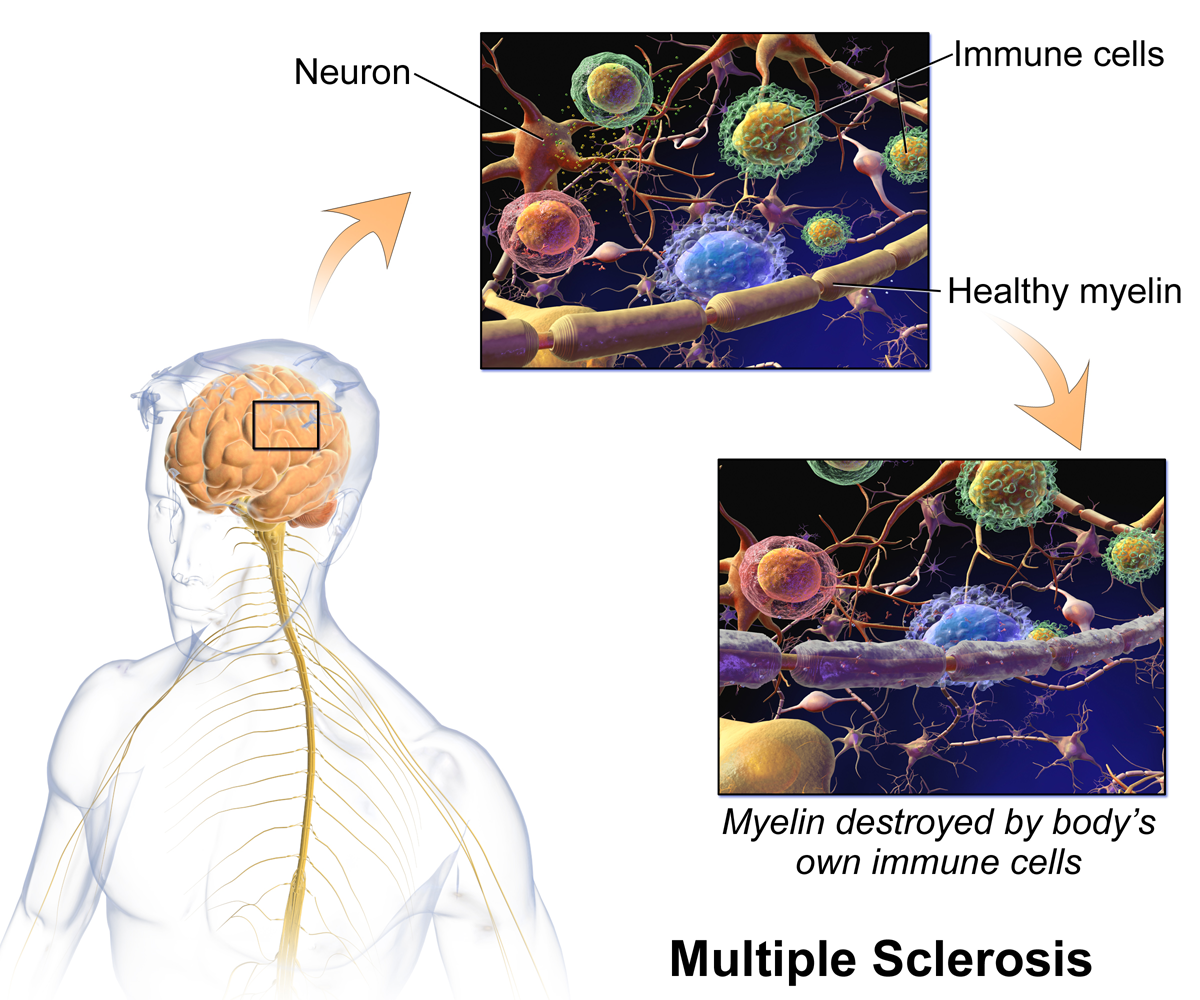
Fig 2: The body's own immune cells destroy myelin in Multiple Sclerosis
© Bruce Blaus
"Multiple sclerosis is caused by the destruction of the myelin sheath of nerve cells, which could be reconstructed by the use of stem cell-based treatment”.
In the past decade, stem cell research has revealed a great potential of utilizing Mesenchymal Stem Cells (MSCs) to reduce inflammation and beneficially modulate the immune system. Apart from the immunosuppressive abilities, MSCs were shown to stimulate the regeneration of a variety of damaged tissues.
The so-called paracrine activity of stem cells has been characterized by the induction of regenerative effects on damaged tissue and neurons. The paracrine action is described by the production of important factors that are secreted by cells, for efficient communication between surrounding cells and tissues. Stem cells themselves have been shown to produce these factors to initiate the communication pathways with the damaged cells and tissues to trigger and stimulate their regeneration (see Fig. 2). The regenerative factors are composed of growth factors, cytokines and cell cycle regulating miRNAs. They are packed in lipid shells (exosomes and microvesicles) for the transport of information, i.e. throughout the body.
New scientific data is constantly emerging, further confirming and elaborating upon the benefits of stem cell-based treatments for neurodegenerative and autoimmune diseases. For more detailed information about the current stem cell research, please click here.
Scientists and physicians are seeing a great potential of stem cells to significantly ameliorate the symptoms MS and to slow down the progression of the disease. In the future it may be possible to completely stop MS from progressing and perhaps even curing the disease, with stem cells or stem cells in combination with other measures. But more clinical work will be necessary to reach the end goal of curing MS.
If you have any questions regarding our stem cell-based treatments for MS, please feel free to contact us or apply for a treatment here.
References and Literature - Stem Cell-based Therapies for Multiple Sclerosis
- Fox, R. J. & Ransohoff, R. M. New directions in MS therapeutics: vehicles of hope. Trends Immunol. 2004; 25(12):632-6.
- Caplan, A. I. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol. 2007; 213(2):341-7.
- Gianvito Martino, Robin J. M. Franklin, Anne Baron Van Evercooren, Douglas A. Kerr. Stem cell transplantation in multiple sclerosis: current status and future prospects. Nat. Rev. Neurol. 2010; 6, 247–255.
- Trapp, B.D. et al. Axonal transection in the lesions of multiple sclerosis. N. Engl. J. Med. 1998; 338, 278–285
- Freedman MS, Bar-Or A, Atkins HL, Karussis D, Frassoni F, Lazarus H, Scolding N, Slavin S, Le Blanc K, Uccelli A; MSCT Study Group. Study Group The therapeutic potential of mesenchymal stem cell transplantation as a treatment for multiple sclerosis: consensus report of the International MSCT Study Group. Multiple Sclerosis. 2010; 16(4) 503–510.
- Karussis D, Karageorgiou C, Vaknin-Dembinsky A, Gowda-Kurkalli B, Gomori JM, Kassis I, Bulte JW, Petrou P, Ben-Hur T, Abramsky O, Slavin S. Safety and Immunological Effects of Mesenchymal Stem Cell Transplantation in Patients With Multiple Sclerosis and Amyotrophic Lateral Sclerosis. Arch Neurol. 2010; 67(10):1187-1194.
- Connick P1, Kolappan M, Crawley C, Webber DJ, Patani R, Michell AW, Du MQ, Luan SL, Altmann DR, Thompson AJ, Compston A, Scott MA, Miller DH, Chandran S. Autologous mesenchymal stem cells for the treatment of secondary progressive multiple sclerosis: an open-label phase 2a proof-of-concept study. Lancet Neurol 2012; 11: 150–56.
- Llufriu S, Sepúlveda M, Blanco Y, Marín P, Moreno B, Berenguer J, Gabilondo I, Martínez-Heras E, Sola-Valls N, Arnaiz JA, Andreu EJ, Fernández B, Bullich S, Sánchez-Dalmau B, Graus F, Villoslada P, Saiz A. Randomized Placebo-Controlled Phase II Trial of Autologous Mesenchymal Stem Cells in Multiple Sclerosis. PLoS One. 2014; 1;9(12).
- Li JF, Zhang DJ, Geng T, Chen L, Huang H, Yin HL, Zhang YZ, Lou JY, Cao B, Wang YL. The Potential of Human Umbilical Cord-Derived Mesenchymal Stem Cells as a Novel Cellular Therapy for Multiple Sclerosis. Cell Transplant. 2014; 23 Suppl 1:S113-22.
- Tyndall A, Walker UA, Cope A, Dazzi F, De Bari C, Fibbe W, Guiducci S, Jones S, Jorgensen C, Le Blanc K, Luyten F, McGonagle D, Martin I, Bocelli-Tyndall C, Pennesi G, Pistoia V, Pitzalis C, Uccelli A, Wulffraat N, Feldmann M. Immunomodulatory properties of mesenchymal stem cells: a review based on an interdisciplinary meeting held at the Kennedy Institute of Rheumatology Division, London, UK, 31 October 2005. Arthritis Research & Therapy 2007; 9:301
- Baraniak PR1, McDevitt TC. Stem cell paracrine actions and tissue regeneration. Regen Med. 2010; 5(1):121-43
- Chandran S, Hunt D, Joannides A, Zhao C, Compston A, Franklin RJ. Myelin repair: the role of stem and precursor cells in multiple sclerosis. Philos Trans R Soc Lond B Biol Sci. 2008; 12;363(1489):171-83.
- Bollini S, Gentili C, Tasso R, Cancedda R. The Regenerative Role of the Fetal and Adult Stem Cell Secretome. J Clin Med. 2013; 17;2(4):302-27
- Yeo RWY, Lai RC, Tan KH Lim SK. Exosome: A Novel and Safer Therapeutic Refinement of Mesenchymal Stem Cell. Exosomes microvesicles. 2013, Vol. 1, 7.
Further References for MSC, BMC, Stemcell Secretome and EVs
- Georg Hansmann, Philippe Chouvarine, Franziska Diekmann, Martin Giera, Markus Ralser, Michael Mülleder, Constantin von Kaisenberg, Harald Bertram, Ekaterina Legchenko & Ralf Hass "Human umbilical cord mesenchymal stem cell-derived treatment of severe pulmonary arterial hypertension". Nature Cardiovascular Research volume 1, pages568–576 (2022).
- Murphy JM, Fink DJ, Hunziker EB, et al. Stem cell therapy in a caprine model of osteoarthritis . Arthritis Rheum. 2003;48:3464–74.
- Lee KB, Hui JH, Song IC, Ardany L, et al. Injectable mesenchymal stem cell therapy for large cartilage defects—a porcine model. Stem Cell. 2007;25:2964–71.
- Saw KY, Hussin P, Loke SC, et al. Articular cartilage regeneration with autologous marrow aspirate and hyaluronic acid: an experimental study in a goat model. Arthroscopy . 2009;25(12):1391–400.
- Black L, Gaynor J, Adams C, et al. Effect of intra-articular injection of autologous adipose-derived mesenchymal stem and regenerative cells on clinical signs of chronic osteoarthritis of the elbow joint in dogs. Vet Ther. 2008;9:192-200.
- Centeno C, Busse D, Kisiday J, et al. Increased knee cartilage volume in degenerative joint disease using percutaneously implanted, autologous mesenchymal stem cells. Pain Physician. 2008;11(3):343–53.
- Centeno C, Kisiday J, Freeman M, et al. Partial regeneration of the human hip via autologous bone marrow nucleated cell transfer: a case study. Pain Physician. 2006;9:253–6.
- Centeno C, Schultz J, Cheever M. Safety and complications reporting on the re-implantation of culture-expanded mesenchymal stem cells using autologous platelet lysate technique. Curr Stem Cell. 2011;5(1):81–93.
- Pak J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose derived stem cells: a case series. J Med Case Rep. 2001;5:296.
- Kuroda R, Ishida K, et al. Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage. 2007;15:226–31.
- Emadedin M, Aghdami N, Taghiyar L, et al. Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch Iran Med. 2012;15(7):422–8.
- Saw KY et al. Articular cartilage regeneration with autologous peripheral blood stem cells versus hyaluronic acid: a randomized controlled trial. Arthroscopy. 2013;29(4):684–94.
- Vangsness CT, Farr J, Boyd J, et al. Adult human mesenchymal stem cells delivered via intra-articular injection to the knee following partial medial meniscectomy. J Bone Joint Surg. 2014;96(2):90–8.
- Freitag, Julien, et al. Mesenchymal stem cell therapy in the treatment of osteoarthritis: reparative pathways, safety and efficacy–a review. BMC musculoskeletal disorders 17.1 (2016): 230.
- Maumus, Marie, Christian Jorgensen, and Danièle Noël. " Mesenchymal stem cells in regenerative medicine applied to rheumatic diseases: role of secretome and exosomes. " Biochimie 95.12 (2013): 2229-2234.
- Dostert, Gabriel, et al. " How do mesenchymal stem cells influence or are influenced by microenvironment through extracellular vesicles communication?. " Frontiers in Cell and Developmental Biology 5 (2017).
- Chaparro, Orlando, and Itali Linero. " Regenerative Medicine: A New Paradigm in Bone Regeneration. " (2016).
- Toh, Wei Seong, et al. " MSC exosome as a cell-free MSC therapy for cartilage regeneration: Implications for osteoarthritis treatment. " Seminars in Cell & Developmental Biology. Academic Press, 2016.
- Chaparro, Orlando, and Itali Linero. " Regenerative Medicine: A New Paradigm in Bone Regeneration. " (2016).
- S. Koelling, J. Kruegel, M. Irmer, J.R. Path, B. Sadowski, X. Miro, et al., Migratory chondrogenic progenitor cells from repair tissue during the later stages of human osteoarthritis , Cell Stem Cell 4 (2009) 324–335.
- B.A. Jones, M. Pei, Synovium-Derived stem cells: a tissue-Specific stem cell for cartilage engineering and regeneration , Tissue Eng. B: Rev. 18 (2012) 301–311.
- W. Ando, J.J. Kutcher, R. Krawetz, A. Sen, N. Nakamura, C.B. Frank, et al., Clonal analysis of synovial fluid stem cells to characterize and identify stable mesenchymal stromal cell/mesenchymal progenitor cell phenotypes in a porcine model: a cell source with enhanced commitment to the chondrogenic lineage, Cytotherapy 16 (2014) 776–788.
- K.B.L. Lee, J.H.P. Hui, I.C. Song, L. Ardany, E.H. Lee, Injectable mesenchymal stem cell therapy for large cartilage defects—a porcine model, Stem Cells 25 (2007) 2964–2971.
- W.-L. Fu, C.-Y. Zhou, J.-K. Yu, A new source of mesenchymal stem cells for articular cartilage repair: mSCs derived from mobilized peripheral blood share similar biological characteristics in vitro and chondrogenesis in vivo as MSCs from bone marrow in a rabbit model , Am. J. Sports Med. 42 (2014) 592–601.
- X. Xie, Y. Wang, C. Zhao, S. Guo, S. Liu, W. Jia, et al., Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration , Biomaterials 33 (2012) 7008–7018.
- E.-R. Chiang, H.-L. Ma, J.-P. Wang, C.-L. Liu, T.-H. Chen, S.-C. Hung, Allogeneic mesenchymal stem cells in combination with hyaluronic acid for the treatment of osteoarthritis in rabbits , PLoS One 11 (2016) e0149835.
- H. Nejadnik, J.H. Hui, E.P. Feng Choong, B.-C. Tai, E.H. Lee, Autologous bone marrow–derived mesenchymal stem cells versus autologous chondrocyte implantation: an observational cohort study , Am. J. Sports Med. 38 (2010) 1110–1116.
- I. Sekiya, T. Muneta, M. Horie, H. Koga, Arthroscopic transplantation of synovial stem cells improves clinical outcomes in knees with cartilage defects , Clin. Orthop. Rel. Res. 473 (2015) 2316–2326.
- Y.S. Kim, Y.J. Choi, Y.G. Koh, Mesenchymal stem cell implantation in knee osteoarthritis: an assessment of the factors influencing clinical outcomes , Am. J. Sports Med. 43 (2015) 2293–2301.
- W.-L. Fu, Y.-F. Ao, X.-Y. Ke, Z.-Z. Zheng, X. Gong, D. Jiang, et al., Repair of large full-thickness cartilage defect by activating endogenous peripheral blood stem cells and autologous periosteum flap transplantation combined with patellofemoral realignment , Knee 21 (2014) 609–612.
- Y.-G. Koh, O.-R. Kwon, Y.-S. Kim, Y.-J. Choi, D.-H. Tak, Adipose-derived mesenchymal stem cells with microfracture versus microfracture alone: 2-year follow-up of a prospective randomized trial , Arthrosc. J. Arthrosc. Relat. Surg. 32 (2016) 97–109.
- T.S. de Windt, L.A. Vonk, I.C.M. Slaper-Cortenbach, M.P.H. van den Broek, R. Nizak, M.H.P. van Rijen, et al., Allogeneic mesenchymal stem cells stimulate cartilage regeneration and are safe for single-Stage cartilage repair in humans upon mixture with recycled autologous chondrons , Stem Cells (2016) (n/a-n/a).
- L. da Silva Meirelles, A.M. Fontes, D.T. Covas, A.I. Caplan, Mechanisms involved in the therapeutic properties of mesenchymal stem cells , Cytokine Growth Factor Rev. 20 (2009) 419–427.
- W.S. Toh, C.B. Foldager, M. Pei, J.H.P. Hui, Advances in mesenchymal stem cell-based strategies for cartilage repair and regeneration , Stem Cell Rev. Rep. 10 (2014) 686–696.
- R.C. Lai, F. Arslan, M.M. Lee, N.S.K. Sze, A. Choo, T.S. Chen, et al., Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury , Stem Cell Res. 4 (2010) 214–222.
- S. Zhang, W.C. Chu, R.C. Lai, S.K. Lim, J.H.P. Hui, W.S. Toh, Exosomes derived from human embryonic mesenchymal stem cells promote osteochondral regeneration, Osteoarthr . Cartil. 24 (2016) 2135–2140.
- S. Zhang, W. Chu, R. Lai, J. Hui, E. Lee, S. Lim, et al., 21 – human mesenchymal stem cell-derived exosomes promote orderly cartilage regeneration in an immunocompetent rat osteochondral defect model , Cytotherapy 18 (2016) S13.
- C.T. Lim, X. Ren, M.H. Afizah, S. Tarigan-Panjaitan, Z. Yang, Y. Wu, et al., Repair of osteochondral defects with rehydrated freeze-dried oligo[poly(ethylene glycol) fumarate] hydrogels seeded with bone marrow mesenchymal stem cells in a porcine model
- A. Gobbi, G. Karnatzikos, S.R. Sankineani, One-step surgery with multipotent stem cells for the treatment of large full-thickness chondral defects of the knee , Am. J. Sports Med. 42 (2014) 648–657.
- A. Gobbi, C. Scotti, G. Karnatzikos, A. Mudhigere, M. Castro, G.M. Peretti, One-step surgery with multipotent stem cells and Hyaluronan-based scaffold for the treatment of full-thickness chondral defects of the knee in patients older than 45 years , Knee Surg. Sports Traumatol. Arthrosc. (2016) 1–8.
- A. Gobbi, G. Karnatzikos, C. Scotti, V. Mahajan, L. Mazzucco, B. Grigolo, One-step cartilage repair with bone marrow aspirate concentrated cells and collagen matrix in full-thickness knee cartilage lesions: results at 2-Year follow-up , Cartilage 2 (2011) 286–299.
- K.L. Wong, K.B.L. Lee, B.C. Tai, P. Law, E.H. Lee, J.H.P. Hui, Injectable cultured bone marrow-derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years’ follow-up , Arthrosc. J. Arthrosc. Relat. Surg. 29 (2013) 2020–2028.
- J.M. Hare, J.E. Fishman, G. Gerstenblith, et al., Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the poseidon randomized trial, JAMA 308 (2012) 2369–2379.
- L. Wu, J.C.H. Leijten, N. Georgi, J.N. Post, C.A. van Blitterswijk, M. Karperien, Trophic effects of mesenchymal stem cells increase chondrocyte proliferation and matrix formation , Tissue Eng. A 17 (2011) 1425–1436.
- L. Wu, H.-J. Prins, M.N. Helder, C.A. van Blitterswijk, M. Karperien, Trophic effects of mesenchymal stem cells in chondrocyte Co-Cultures are independent of culture conditions and cell sources , Tissue Eng. A 18 (2012) 1542–1551.
- S.K. Sze, D.P.V. de Kleijn, R.C. Lai, E. Khia Way Tan, H. Zhao, K.S. Yeo, et al., Elucidating the secretion proteome of human embryonic stem cell-derived mesenchymal stem cells , Mol. Cell. Proteomics 6 (2007) 1680–1689.
- M.B. Murphy, K. Moncivais, A.I. Caplan, Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine , Exp. Mol. Med. 45 (2013) e54.
- M.J. Lee, J. Kim, M.Y. Kim, Y.-S. Bae, S.H. Ryu, T.G. Lee, et al., Proteomic analysis of tumor necrosis factor--induced secretome of human adipose tissue-derived mesenchymal stem cells , J. Proteome Res. 9 (2010) 1754–1762.
- S. Bruno, C. Grange, M.C. Deregibus, R.A. Calogero, S. Saviozzi, F. Collino, et al., Mesenchymal stem cell-derived microvesicles protect against acute tubular injury, J. Am. Soc. Nephrol. 20 (2009) 1053–1067.
- M. Yá˜nez-Mó, P.R.-M. Siljander, Z. Andreu, A.B. Zavec, F.E. Borràs, E.I. Buzas, et al. Biological properties of extracellular vesicles and their physiological functions (2015).
- C. Lawson, J.M. Vicencio, D.M. Yellon, S.M. Davidson, Microvesicles and exosomes: new players in metabolic and cardiovascular disease , J. Endocrinol. 228 (2016) R57–R71.
- A.G. Thompson, E. Gray, S.M. Heman-Ackah, I. Mager, K. Talbot, S.E. Andaloussi, et al., Extracellular vesicles in neurodegenerative diseas—pathogenesis to biomarkers, Nat. Rev. Neurol. 12 (2016) 346–357.
- I.E.M. Bank, L. Timmers, C.M. Gijsberts, Y.-N. Zhang, A. Mosterd, J.-W. Wang, et al., The diagnostic and prognostic potential of plasma extracellular vesicles for cardiovascular disease , Expert Rev. Mol. Diagn. 15 (2015) 1577–1588.
- T. Kato, S. Miyaki, H. Ishitobi, Y. Nakamura, T. Nakasa, M.K. Lotz, et al., Exosomes from IL-1 stimulated synovial fibroblasts induce osteoarthritic changes in articular chondrocytes , Arthritis. Res. Ther. 16 (2014) 1–11.
- R.W.Y. Yeo, S.K. Lim, Exosomes and their therapeutic applications, in: C. Gunther, A. Hauser, R. Huss (Eds.), Advances in Pharmaceutical Cell TherapyPrinciples of Cell-Based Biopharmaceuticals, World Scientific, Singapore, 2015, pp. 477–491.
- X. Qi, J. Zhang, H. Yuan, Z. Xu, Q. Li, X. Niu, et al., Exosomes secreted by human-Induced pluripotent stem cell-derived mesenchymal stem cells repair critical-sized bone defects through enhanced angiogenesis and osteogenesis in osteoporotic rats , Int. J. Biol. Sci. 12 (2016) 836–849.
- R.C. Lai, F. Arslan, S.S. Tan, B. Tan, A. Choo, M.M. Lee, et al., Derivation and characterization of human fetal MSCs: an alternative cell source for large-scale production of cardioprotective microparticles , J. Mol. Cell. Cardiol. 48 (2010) 1215–1224.
- Y. Zhou, H. Xu, W. Xu, B. Wang, H. Wu, Y. Tao, et al., Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro , Stem Cell Res. Ther. 4 (2013) 1–13.
- Y. Qin, L. Wang, Z. Gao, G. Chen, C. Zhang, Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo , Sci. Rep. 6 (2016) 21961.
- M. Nakano, K. Nagaishi, N. Konari, Y. Saito, T. Chikenji, Y. Mizue, et al., Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by exosome transfer into damaged neurons and astrocytes , Sci. Rep. 6 (2016) 24805.
- K. Nagaishi, Y. Mizue, T. Chikenji, M. Otani, M. Nakano, N. Konari, et al., Mesenchymal stem cell therapy ameliorates diabetic nephropathy via the paracrine effect of renal trophic factors including exosomes , Sci. Rep. 6 (2016) 34842.
- S.R. Baglio, K. Rooijers, D. Koppers-Lalic, F.J. Verweij, M. Pérez Lanzón, N. Zini, et al., Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species , Stem Cell Res. Ther. 6 (2015) 1–20.
- T. Chen, R. Yeo, F. Arslan, Y. Yin, S. Tan, Efficiency of exosome production correlates inversely with the developmental maturity of MSC donor, J. Stem Cell Res. Ther. 3 (2013) 2.
- R.C. Lai, S.S. Tan, B.J. Teh, S.K. Sze, F. Arslan, D.P. de Kleijn, et al., Proteolytic potential of the MSC exosome proteome: implications for an exosome-mediated delivery of therapeutic proteasome , Int. J. Proteomics 2012 (2012) 971907.
- T.S. Chen, R.C. Lai, M.M. Lee, A.B.H. Choo, C.N. Lee, S.K. Lim, Mesenchymal stem cell secretes microparticles enriched in pre-microRNAs , Nucleic Acids Res. 38 (2010) 215–224.
- R.W. Yeo, R.C. Lai, K.H. Tan, S.K. Lim, Exosome: a novel and safer therapeutic refinement of mesenchymal stem cell, J. Circ. Biomark. 1 (2013) 7.
- R.C. Lai, R.W. Yeo, S.K. Lim, Mesenchymal stem cell exosomes, Semin. Cell Dev. Biol. 40 (2015) 82–88.
- B. Zhang, R.W. Yeo, K.H. Tan, S.K. Lim, Focus on extracellular vesicles: therapeutic potential of stem cell-derived extracellular vesicles , Int. J. Mol. Sci. 17 (2016) 174.
- Hu G-w, Q. Li, X. Niu, B. Hu, J. Liu, Zhou S-m, et al., Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice , Stem Cell Res. Ther. 6 (2015) 1–15.
- J. Zhang, J. Guan, X. Niu, G. Hu, S. Guo, Q. Li, et al., Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis , J. Transl. Med. 13 (2015) 1–14.
- B. Zhang, M. Wang, A. Gong, X. Zhang, X. Wu, Y. Zhu, et al., HucMSC-exosome mediated-Wnt4 signaling is required for cutaneous wound healing, Stem Cells 33 (2015) 2158–2168.
- B. Zhang, Y. Yin, R.C. Lai, S.S. Tan, A.B.H. Choo, S.K. Lim, Mesenchymal stem cells secrete immunologically active exosomes , Stem Cells Dev. 23 (2013) 1233–1244.
- C.Y. Tan, R.C. Lai, W. Wong, Y.Y. Dan, S.-K. Lim, H.K. Ho, Mesenchymal stem cell-derived exosomes promote hepatic regeneration in drug-induced liver injury models , Stem Cell Res. Ther. 5 (2014) 1–14.
- C. Lee, S.A. Mitsialis, M. Aslam, S.H. Vitali, E. Vergadi, G. Konstantinou, et al., Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia-induced pulmonary hypertension , Circulation 126 (2012) 2601–2611.
- B. Yu, H. Shao, C. Su, Y. Jiang, X. Chen, L. Bai, et al., Exosomes derived from MSCs ameliorate retinal laser injury partially by inhibition of MCP-1 , Sci. Rep. 6 (2016) 34562.
- Jo CH, Lee YG, Shin WH, et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof of concept clinical trial. Stem Cells. 2014;32(5):1254–66.
- Vega, Aurelio, et al. Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation. 2015;99(8):1681–90.
- Davatchi F, Sadeghi-Abdollahi B, Mohyeddin M, et al. Mesenchymal stem cell therapy for knee osteoarthritis. Preliminary report of four patients. Int J Rheum Dis. 2011;14(2):211–5
- Hernigou P, Flouzat Lachaniette CH, Delambre J, et al. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case- controlled study. Int Orthop. 2014;38(9):1811–1818
- Galli D, Vitale M, Vaccarezza M. Bone marrow-derived mesenchymal cell differentiation toward myogenic lineages: facts and perspectives. Biomed Res Int. 2014;2014:6.
- Beitzel K, Solovyova O, Cote MP, et al. The future role of mesenchymal Stem cells in The management of shoulder disorders . Arthroscopy. 2013;29(10):1702–1711.
- Isaac C, Gharaibeh B, Witt M, Wright VJ, Huard J. Biologic approaches to enhance rotator cuff healing after injury. J Shoulder Elbow Surg. 2012;21(2):181–190.
- Malda, Jos, et al. " Extracellular vesicles [mdash] new tool for joint repair and regeneration. " Nature Reviews Rheumatology (2016).
Further References about PRP
- Rubio-Azpeitia E, Andia I. Partnership between platelet-rich plasma and mesenchymal stem cells: in vitro experience. Muscles Ligaments Tendons J. 2014;4(1):52–62.
Extras
- Xu, Ming, et al. " Transplanted senescent cells induce an osteoarthritis-like condition in mice. " The Journals of Gerontology Series A: Biological Sciences and Medical Sciences (2016): glw154.
- McCulloch, Kendal, Gary J. Litherland, and Taranjit Singh Rai. " Cellular senescence in osteoarthritis pathology ." Aging Cell (2017).
Contraindications
Our stem cell treatments are experimental, but we only treat patients for whom we believe the risk/benefit ratio indicates treatment based on the state of the art, i.e., medical, scientific evidence.
Please understand that we therefore do not treat patients for whom the following points apply:
- Active cancer in the last two years
- Not yet of legal age
- Existing pregnancy or lactation period
- Unable to breathe on own, ventilator
- Difficulty breathing in supine position
- Dysphagia (extreme difficulty swallowing)
- Psychiatric disorder
- Active infectious disease (Hepatitis A, B, C, HIV, Syphilis, or other)