Stem Cell Therapy for Back Pain, Spine and Cervical Conditions at
ANOVA IRM in Offenbach, Germany
Back pain affects almost all of us as soon as we reach a sufficient age, especially when healthy physical activity is missing or excessive extreme sport challenges your spine. According to NINDS (National Institute of Neurological Disorders and Stroke), 80% of all adults suffer from back pain at least temporarily. In general, back pain represents one of the most common reasons why people see a doctor, and additionally, it is one of the leading causes of disability all over the world, as reported by the Global Burden of Disease Study (GBD).
Back pain can arise from false postures, sitting at a computer the wrong way, sports accidents or just aeging and wear of discs. In most cases of back pain, the physiological cause is a intervertebral disc condition or degenerative disc disease.
While stem cell-based treatments are a cornerstone of our approach for inflammatory and degeneration-induced back pain, ANOVA IRM also integrates other advanced diagnostic and minimally invasive therapeutic options. Our goal is to provide a comprehensive, individualized plan based on precise identification of your pain source, which may include stem cells, targeted nerve therapies, or other innovative strategies to help you find relief and improve function.
Back Pain - Diagnostics - Treatment - Stem cells - Medication
On this page we inform you about back pain, degenerative disc disease, spinal and vertebral conditions covering an overview on important aspects of causes, treatment options, precision diagnostics that reveal the cause of pain and location of the pain-causing defect, as well as our stem cell-based therapies that we offer in Offenbach (near Frankfurt am Main airport) Germany.
Jump directly to the following topics:
- Conventional therapies
- ANOVA therapies for e.g. back pian, degenerative disc disease
- Expectations and limits
- Avoid disc replacement
- Our treatment options
- Workflow of the treatment process
- Diagnostics of pain-causing defects
- The ANOVA difference: targeted treatment
- You want a second opinion
- FAQ- frequently-asked questions
- Sources and Literature
- Our Advanced Diagnostic Approach
- Minimally Invasive Nerve Ablation
- Supportive Senolytic Therapy
We Treat the Following Back Pain Conditions:
- Back pain
- Degenerative disc disease
- Disc-Prolaps
- Degenerative chnages of discs
- Injuries of the thoracic and lumbar spine
- Injuries of the thoracic and lumbar spinal chord
- Spinal cord transections (partial transections and, in combination with other therapies, complete but responsive transections)
- Chronic pain and inflammation of the spine
- Facet Joint Syndrome / Facet Arthropathy
- Pain related to Modic Changes (suitable for BVN Ablation)
Conventional Treatments for Back Pain and Degenerative Disc Disease
Back pain and spinal conditions are usually treated with physiotherapy, medications and injections that target the painful symptoms. When this mere treatment of symptoms fails to allow for a bareable pain level, invasive methods, such as spinal and back operations (surgeries) are usually the only remaining option. Lumbar spinal fusion is a conventional therapy that fixes vertebrae and attaches them to one another, reducing pain but also mobility. Disc prostheses replace damaged discs and thereby maintaining motion of the spinal segment and reducing pain but usually not completely eliminating pain.
Both surgery and pain relief medications seldom satisfy the patient, as shown by statistics conducted in clinical institutions. This does not come as a surprise, because none of these options offer any real solution to the root of the problem. While these common treatments can offer improvements (and are sometimes even necessary), the choice of the correct treatment option depends on the origin and the severity of the pain. Is a surgical procedure necessary? And if yes, what can patients expect as an outcome?
In general, before the decision for getting an operation for back pain relief is considered, all other methods should be carefully evaluated. Intensive diagnostics should be applied to identify the cause of pain. An exception to this rule is when there is an immediate need for a surgical intervention, for instance in cases with entire loss of control in the leg(s). However, back pain operations are not at all a 100% guarantee that the patient will fully recover and live a pain-free life – quite often the opposite result happens. More on conventional options is summarized by Lumitex in a spinal surgery overview. A comprehensive list of surgery risks has been compended by Johns Hopkins exemplified for lumbar fusion.
Stem Cell Treatments for Back Pain and Degenerative Disc Disease at
ANOVA Institute for Regenerative Medicine - Offenbach, Germany
BMC, Secretome/Exosomes, PRP
Potency Hypothesis of Stem Cell Therapies
Stem cells possess the potential to communicate with the immune cells that elicit the inflammation and by natural, so far not understood mechanisms may inhibit this immune-over-reaction. Furthermore, stem cells have the ability to stimulate regeneration of tissue thereby counteracting the wear and loss of cartilage in the affected joints. Negative long-term effects similar to cortisone are not expected. The aim of a stem cell treatment is therefore, the fast relief of pain, the slowing of the disease progression and in the best cases to even support joint and disc regeneration. This can dramatically increase the quality of life, especially for patients with severe pain, as well as the movement duration and range.
Is it Possible to Relief Pain and Avoid Disc Replacement?
Yes, there are different innovative (experimental) treatment concepts which specifically target cartilage and disc defects with stem cell therapies. Combined with physiotherapy, they help to avoid disc replacement also in the long term. A surgical disc replacement requires long-lasting rehabilitation and often offers only moderate treatment success. Patients often re-acquire their strength and mobility after months but fail to improve their level of activity. Additionally, disc implants have to be replaced regularly which results in several surgical interferences over the years for younger patients.
A specific, well-tailored stem cell therapy in contrast could postpone the surgical procedure and might even help to avoid disc replacement. For this, autologous (patient's own) stem cells are used to halt inflammation and initiate the body's own regeneration and repair system. New evidence-based external scientific and clinical studies have shown that stem cell-based therapies are able to modulate immune responses in spinal conditions and to stimulate regenerative processes in bone, disc and cartilage by e.g. stimulation of chondrocyte replacement (the cells that produce cartilage).
Avoid joint disc replacement surgery
ANOVA IRM - Germany
Two Targeted Effects: Pain Relief and Progression Improvement
A stem cell treatment can elicit two effects in affected discs, joints and vertebrae that build on one another. First, due to the modulation of the underlying immune reaction the stem cell injection inhibits the inflammation. As inflammation often is the main cause of pain, targeted stem cell injection often results in immediate pain relief up to complete pain-free motion of the treated joint.
Second, the effects on regeneration build on the inhibition of inflammation. The joint, dics and nerves return to a resting-phase and are now able to react to healing and regenerative stimuli. With adequate on-going stem cell therapy in combination with e.g. physiotherapy, regeneration of cartilage, discs and bone can take place. As all effects are patient- and disease stage-dependent and may be influenced by additional, external factors, we always apply individual treatment plans. In general, pain is relieved, inflammation is reduced, further degradation and wear of the spinal system is slowed up to regeneration of discs and cartilage.
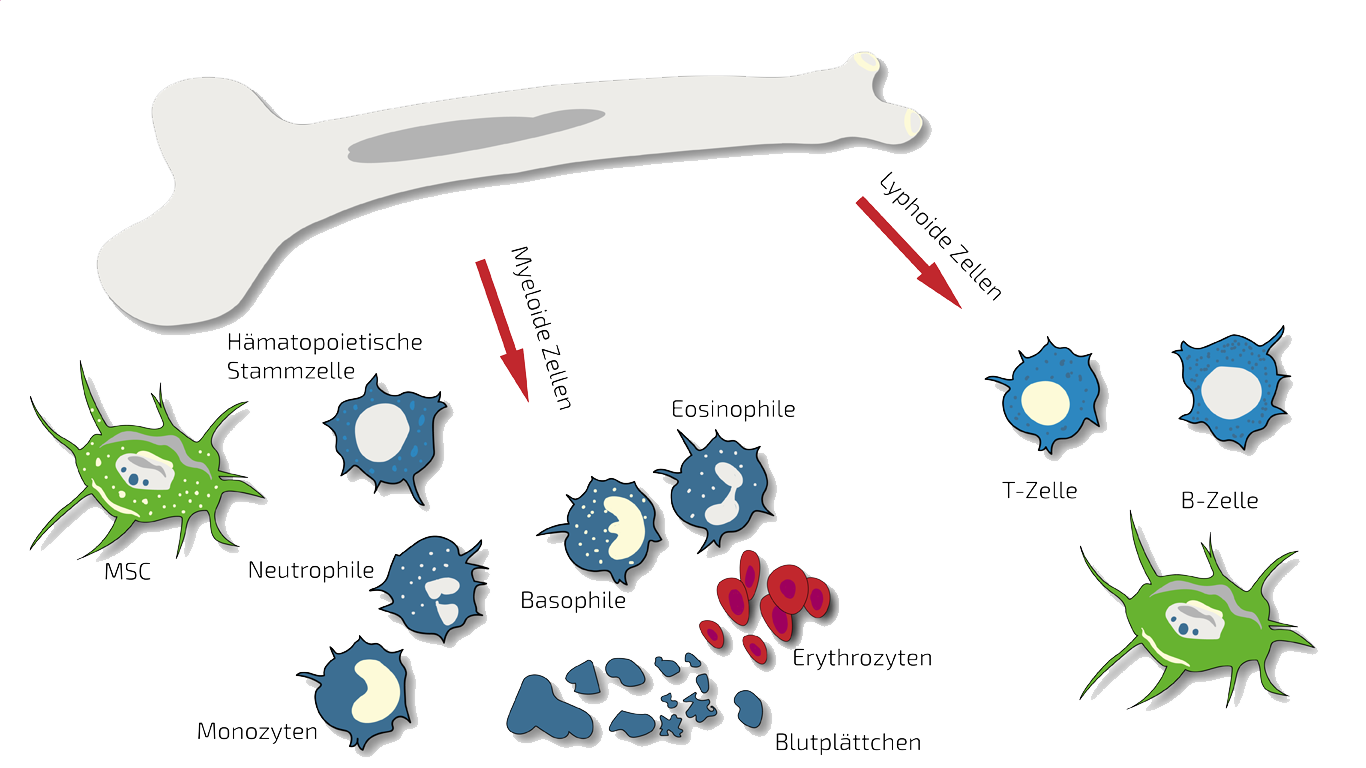
BMC - Bone Marrow Concentrate - Autologous
Autologous (self) BMC are our main therapy option for locally-restricted and mild conditions as BMC is a one bone marrow donation - one injection treatment.
In such cases we treat specifically this disc or this spinal segment with targeted, localized BMC injections. BMC contains autologous meaning patients own, adult stem cells (hematopoietic and mesenchymal stem cells in natural composition) which we isolate and concentrate from your pelvis crest in a short process under slight sedation.
These stem cells are supposed to inhibit the inflammation thereby relieving you from pain and to stimulate regeneration of the spinal segment. For an on-going therapy, we combine BMC with PRP (platelet-rich plasma) or MSEC (see below). More information about this type of stem cell therapy is summarized on our page an BMC.
PRP - Platelet-Rich Plasma - Autologous
PRP is a comparably inexpensive experimental therapy as platelets (thrombocytes naturally containing growth factors and stimulants) are isolated from autologous (own) blood without isolation of stem cells.
For spinal conditions, we often use PRP in combination with BMC and PRP is administered in-between BMC treatments to continuously support the regeneration-stimulating effect. Besides this, PRP is well-known as a stimulant for wound healing in e.g. periodontitis therapy or as a measure against hair loss. More on PRP (as a combination therapy) is summarized on our PRP overview page.
MSEC - Mesenchymal Stem Cell Secretome - Exosomes - Autologous
In later stages or more wide-spread damage, we treat patients with spinal conditions with MSEC (Secretome, Exosomen, EVs) of mesenchymal stem cells (MSC, AD-MSC, adipose-derived, fat-derived stem cells) which we harvest from the patients belly in a mini-liposuction (very brief and limited liposuction) under slight sedation. Worldwide, ANOVA is the first stem cell clinic to acquire legal permission form the responsible governmental authorities and therefore, offers high quality, safe and legally-controlled autologous (own) exosome-containing secretome.
The main advantage of MSEC is that in contrast to live stem cells which would loose their therapeutic potency, can be frozen without loss of exosomes. This enables us to produce 10-20 injection doses from one liposuction which can then be administered over a longer treatment period. This is especially advantageous for serious cases of wide-spread Polyarthritis. What a Secretome/Exosome is and how they compare is explained on our overview page.
Hyaluronic Acid - HA
Another, cost efficient supportive treatment is injection of hyaluronic acid into affected discs.
Especially in early cases, this can reduce pain and result in a better "buffering" of the spinal segment.
Hyaluronic acid alone, on the other hand, is not expected to have regenerative effects on the damaged disc, cartilage or bone.
Therefore, we usually combine HA with BMC treatment and HA is given intermittendly between BMC treatments.

Hyaluronic acid - HA
ANOVA IRM - Germany
Minimally Invasive Nerve Ablation (RFA): Targeted Pain Relief
For specific types of chronic back pain where diagnostic investigations (including advanced MRI and potentially diagnostic blocks) point to the facet joints or the basivertebral nerve as the primary pain generators, Radiofrequency Ablation (RFA) offers a highly effective, minimally invasive treatment. This image-guided procedure uses targeted heat to deactivate the small nerves transmitting pain signals from these structures. RFA can provide significant and lasting pain relief for conditions like facet joint syndrome or certain types of vertebrogenic low back pain (e.g., related to Modic changes), often allowing patients to avoid or delay more invasive surgical options.
Supportive Senolytic Therapy: Addressing Cellular Aging
Recent scientific research highlights the role of senescent (aging) cells in promoting degenerative joint disease and intervertebral disc degeneration. These cells accumulate with age and in areas of chronic injury, releasing inflammatory factors that can exacerbate pain and hinder repair. Senolytic therapies are designed to selectively clear these senescent cells. At ANOVA IRM, we may discuss the potential for concomitant senolytic therapy as a supportive measure to complement our primary treatments, aiming to reduce the overall inflammatory burden and create a more favorable environment for healing and regeneration.
Contraindications
Our stem cell treatments are experimental, but we only treat patients for whom we believe the risk/benefit ratio indicates treatment based on the state of the art, i.e., medical, scientific evidence.
Please understand that we therefore do not treat patients for whom the following points apply:
- Active cancer in the last two years
- Not yet of legal age
- Existing pregnancy or lactation period
- Unable to breathe on own, ventilator
- Difficulty breathing in supine position
- Dysphagia (extreme difficulty swallowing)
- Psychiatric disorder
- Active infectious disease (Hepatitis A, B, C, HIV, Syphilis, or other)
Therapy Workflow for Spinal Conditions
The precise workflow is described in detail on the stem cell- specific pages of BMC, Secretome/Exosomes und PRP (as combination therapy).
All therapies are divided into phases such as evaluation of the medical history (we analyze your current therapies and medical records), initial counselling and evaluation of potential, patient-individual benefit of a stem cell therapy (indication statement), preliminary examinations, diagnostics, consultation on all therapy options, preparation of an individual treatment plan including cost estimate, which will outline the recommended approach, potentially including **stem cell therapy (BMC/MSEC), minimally invasive nerve ablation, supportive senolytic therapy, or a combination**, based on your specific diagnostic findings. This is followed by the procedure itself (e.g., tissue harvesting, stem cell production/application, ablation procedure), including quality control where applicable.
There are two special features for patients with spinal conditions or degenerative disc disease: If your previous diagnostics have not found the specific causes of your spinal pain, we will ensure you have the necessary precise imaging. This often includes **MRI with intravenous contrast agent** to pinpoint active inflammation – a key indicator of the pain source – and specialized views like **sagittal oblique sequences for the cervical spine**, if indicated.
Unfortunately, according to the risk-benefit ratio, we cannot treat children or pregnant women. In addition, other factors can also be exclusion criteria.
How Long Does a Stem Cell Therapy Take?
The initial analyses and counselling can be done without you having to travel to Offenbach (near Frankfurt/Main, Germany). This period can be 2 weeks up to months depending on the availability of patients slots. If you live further away, we will conduct the initial discussions by telephone or video conference. For the actual treatment, you will travel to Offenbach. Then, depending on the therapy, the tissue collection, quality control and treatment type it will take as follows:
BMC- und PRP-therapy
Each donation and application of BMC and PRP (as combination therapy) on-site period: 2 days (consecutive days).
Secretome/Exosome-therapy:
Preparation and harvest of the fat (mini-liposuction) need once 2 days (consecutive days) in Offenbach, followed by enrichment of the mesenchymal stem cells (Secretome/Exosome) and quality control. Approximately 4 weeks after the isolation, the therapy begins according to the therapy plan determined with you. You will then come to Offenbach am Main (Germany) several times for the application. The shelf life of the secretome (exosomes) is 2 years.
How Much Does Stem Cell Treatment Cost?
Our treatments are always tailored to your specific situation, disease, stage and other factors. The therapies differ in the product used (BMC, secretome, PRP or hyaluronic acid), the frequency of treatment as well as the further examinations and your sedation and anesthesia wishes. A treatment for osteoarthritis and arthritis can cost from a few hundred to several thousand euros. You will receive a cost estimate for all treatments in advance so that you can accurately estimate what a treatment would cost in your individual case.
Does my Health Insurance Cover the Therapy Costs?
Unfortunately, at the moment it is assumed that health insurance companies do not cover the costs of experimental therapies (BMC, secretome, PRP, micro-fracture technique), i.e. you will have to bear the costs entirely yourself.
How Does the ANOVA Therapy differ?
Diagnostics – We Look for the Cause of your Pain
Dr. mult. Michael K. Stehling, the founder of ANOVA IRM and the Vitus Prostate Center , is a radiologist (MD) and holds a PhD in physics. For this reason, the ANOVA Institute for Regenerative Medicine, in cooperation with the Prof. Stehling Institute for Diagnostic Imaging located in the same building, has the capability to use special precision diagnostics such as high-resolution MRI, often performed with an **intravenous contrast agent** to visualize inflammation, and specialized sequences like **sagittal oblique views for the cervical spine**.
Compared to standard MRIs which primarily show anatomy, **contrast-enhanced MRI** is crucial because it can highlight areas of active inflammation, directly indicating the likely source of pain. Similarly, **sagittal oblique views** are vital for accurately assessing nerve pathways in the neck. This level of diagnostic detail allows for highly targeted treatment. This enables us to determine individually how patients should be treated and where the stem cells should be applied.
Typical radiographic findings of DDD include disc space narrowing, displacement of vertebral bodies, fusion of adjacent vertebral bodies, and development of bone spurs (osteophytes) originating from the affected vertebral bodies. MRI affords a grading of the degenerative process: the Pfirrmann scale. It ranges from a grade I, healthy, homogeneously white disc of normal height to a grade V almost completely collapsed black and dehydrated disc.
In order to correctly assess secondary changes,For instance, high-resolution MRI incorporating **sagittal oblique views** to clearly depict the neuroforamina in the cervical spine, and sequences performed **after the injection of intravenous contrast agent** to reveal inflammatory changes, are critical for accurate assessment but not always standard elsewhere. Because few radiology departments routinely perform these specific sequences, we often recommend patients have them taken here during their appointment. At ANOVA, we have developed specific imaging protocols for degenerative disc and spine conditions to obtain all the relevant information to advise patients what kind of treatment is best for them and what results they can expect from it.
Furthermore, in consultation with you, we supplement our patient-specific diagnostics with specific blood tests on hormones, inflammation parameters and other factors that are important in your case, or recommend further examinations such as a preventive MRI spinal scan.
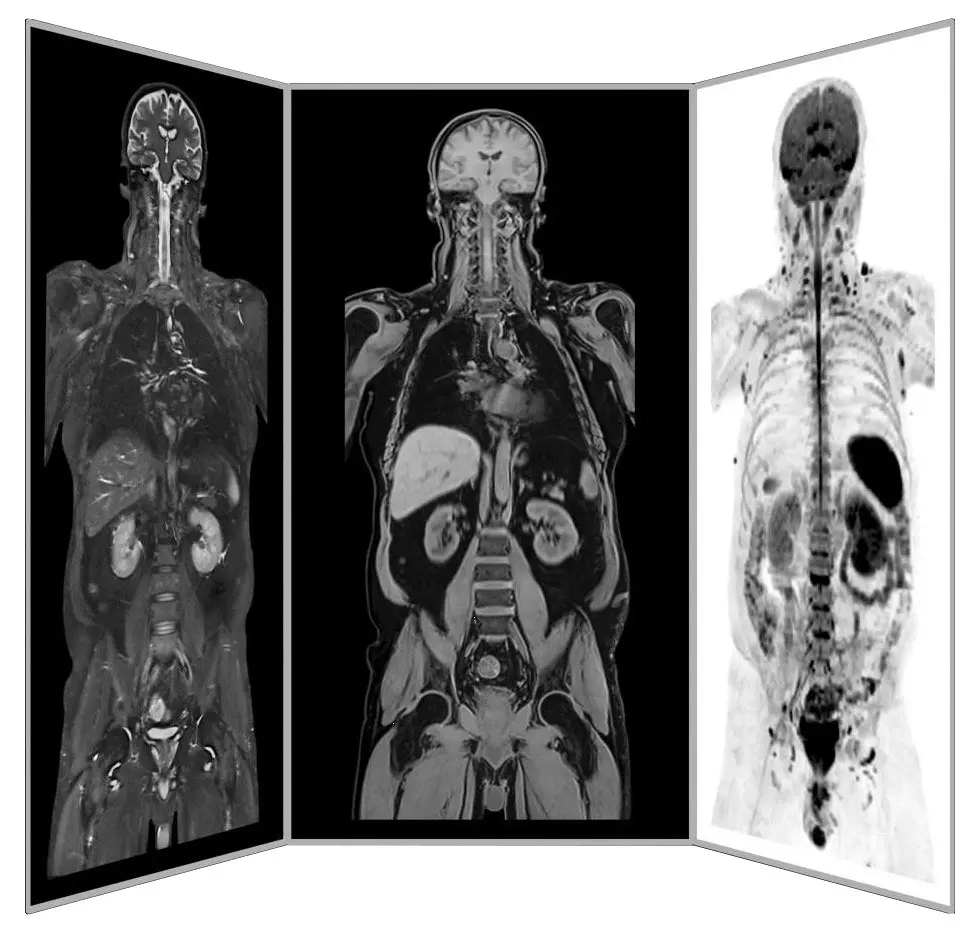
Precision MRI scans - find the source of pain
ANOVA IRM © Siemens Healthcare GmbH
How Does the ANOVA Therapy Differ?
We Implant the Stem Cells Precisely Where They are Needed
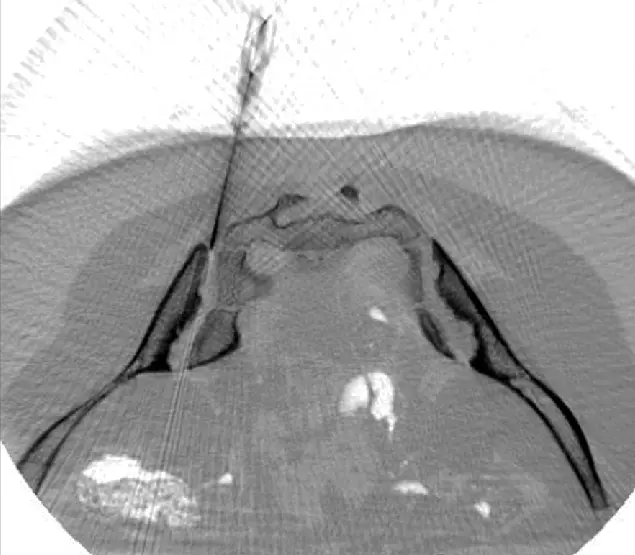
CT-assisted stem cell injection into joints
ANOVA IRM © Siemens Healthcare GmbH
Based on our specific diagnostics using arthro-MRI and non-radioactive contrast medium MRIs, we can, in contrast to many other clinics, deliver therapies like **stem cells (BMC) or nerve ablation probes** with image support (e.g., using CT or Fluoroscopy) precisely to the affected area (joint, disc, specific nerve). This means we can inject into and at joints, discs and vertebrae to specifically and quickly trigger an effect where inflammation causes pain. All interventions are perfomed under supervision and care of our anesthesiologist and are pain free.
A purely intravenous administration, as many other clinics do, is only performed for the secretome (exosomes) if this is to be used additionally as a supportive or preventive measure because joint problems are present in several places in the body as the secretome is aimed to centrally modulate the immune response in order to inhibit over-reactions.
Of course, we will thoroughly advise you in the early process and the on-site consultation in advance on all steps and discuss alternatives and expectations.
Are you Interested but Uncertain? Book a Counselling Appointment!
Our patient care managers are happy to inform you about what information we need upfront (MRI, CT, X-ray), how to transfer large data files and schedule a counseling appointment with our physicians for you. Our patient care managers are happy to inform you about what information we need upfront, how to transfer large data files and schedule a counseling appointment with our physicians for you. Please use our contact form to support a fast processing of your case and request.
You are also always welcome to send us an e-mail about your case. The counseling appointment may also take place per telephone or video chat if you live outside Germany. For more intense counseling or additional diagnostic evaluations you may also book an on-site appointment. We can perform needed MRI on the same day. All services rendered by our patient care team are free of charge and we inform you about all physician appointment charges up-front.

Avoid joint replacement with stem cell treatment
ANOVA IRM - Germany
External Scientific Results:
Stem Cells are Promising Therapy Option that Work on a Cellular Level
In animal models of disc degeneration, MSCs have shown to restore normal disc structure. Since disc degeneration seems to depend on alterations of nucleus pulposus (NP) cells, Strassburg and colleagues have investigated the interactions between MSCs and degenerated NP cells, in greater detail.
Similar to other degenerative diseases where stem cell therapies have been proven to be successful (i.e. neurodegenerative diseases), they found that the two cell types primarily communicate via an extensive direct transfer of membrane components and extracellular vesicles, which is known as the Stem Cell Secretome.
Stem cells can trigger regenerative effects by improving the problems at the root, where the catabolic microenvironment causes bad disc quality due to minimal cell proliferation. Stem cells can help with their intrinsic inter-cellular communication factors, the so called paracrine factors, also known as the Stem Cell Secretome. The ANOVA Stem Cell Secretome therapy is designed to harness and concentrate these factors.
ANOVA Institute for Regenerative Medicine has moved long past a one-fits-all stem cell therapy. Different cell therapies have been proven to be efficient for different back pain and spinal conditions, syndromes and degenerations. All of these, namingly Bone Marrow Concentrate (BMC), Mesenchymal Stem Cell Secretome (MSEC), Platelet Rich Plasma (PRP) and tissue transplants grown from stem cells have shown to induce regenerative effects for different orthopedic pathologies.
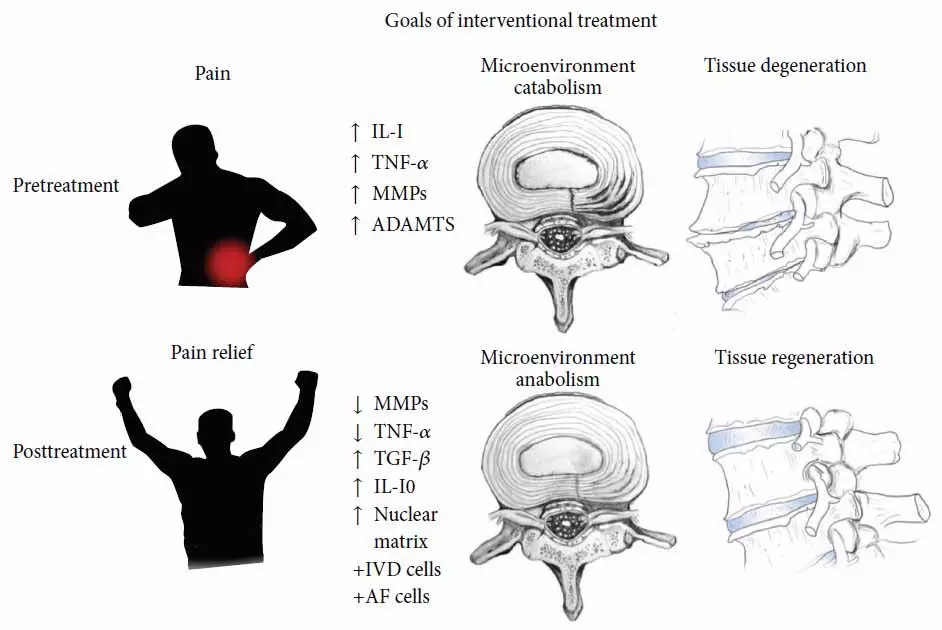
Figure: Schematic illustration of discogenic degenerative diseases of the disc – one of the most frequent sources of back pain.
ANOVA offers these treatments in a personalized treatment plan, based on your current health status. As with any type of treatment, experimental therapy such as stem cell-based therapy cannot promise any success. Before the attending physician can suggest an experimental therapy, they will individually assess whether the benefits of the therapy are given for the patient and make sure they outweigh the potential risks. With this approach, ANOVA establishes a safe method to offer cellular regenerative treatment for back pain and spinal conditions in cases where it is beneficial. Unmatched quality worldwide and developed by a team of German scientists and medical professionals in a dedicated private German institute.
If you are interested to apply for a stem cell-based treatment for back pain, of wish to know more information, please feel free to contacts us.
Frequently Asked Questions:
Stem Cell Treatments and Regenerative Therapies for Spinal Conditions
How Does a Disc Compression Cause Numbness or Pain
As we grow older, our intervertebral disc grow old with us and loose their flexibility. The cumulative stress induced by wrong body positioning, lack of healthy motion and often under-nurishing due to lack of passive motion of the discs leads to a slow narrowing of the intervertebral space and shrinking of the discs.
This can lead to the situation that parts of the intervertebral dics are pushed outwards in regions where this is possible. In some cases this is a region where a exits the spinal chord to innervate muscles of that region. This can result in moderate pain during motion which might with time develop into excruciating pain.
In addition, when spinal discs are then compressed either too long or strongly by a sudden movement, the disc can rupture and release parts of its inner gel-like contents. This might elicit an inflammatory reaction which sppeds up the degenerative process.
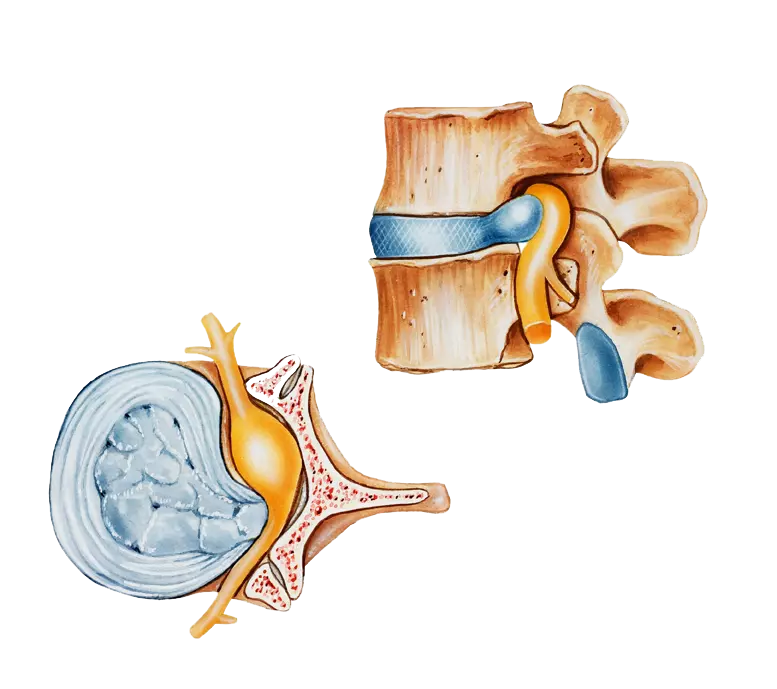
Painful nerve compression due to disc prolaps
ANOVA IRM - Germany
How Does Cortisone Work?
Injection of cortisone (corticosteroids) into inflamed joints is still widely used today. It typically results in rapid pain relief, but its effects usually do not last longer than 4 weeks. However, in the long run, cortisone injections accelerate joint wear by damaging cartilage cells, which are essential for maintaining articular cartilage.
What are NSAID, NSAP, NSAR?
All these abbreviations stand for anti-inflammatory drug groups that are used for inflammatory processes and diseases such as rheumatism, osteoarthritis and arthritis. NS always stands for non-steroidal, i.e. substances that are not derived from steroids such as cortisone. All non-steroidal anti-inflammatory drugs have sometimes serious side effects such as damage to the gastrointestinal mucosa or heart and kidney damage. Some preparations have therefore been withdrawn from the market.
- NSAID - non-steroidal anti-inflammatory drug
- NSAP - non-steroidal anti-inflammatory drug
- NSAID - non-steroidal anti-inflammatory drug (translated non-steroidal anti-inflammatory drug)
What Are Anti-Phlogistic Drugs?
Anti-phlogistic drugs are anti-inflammatory drugs. Anti-inflammatory drugs include the following groups of drugs:
- Glucocorticoids (e.g., cortisone).
- Non-steroidal anti-inflammatory drugs (NSAIDs, non-steroidal anti-rheumatic drugs)
- Immunosuppressants (DMARDs, disease-modifying anti-rheumatic drugs
- Certain novel biologic-derived drugs (biologicals, e.g., JAK inhibitors)
Effects, Risks and Side Effects of Drugs and Treatments
Patients are always individuals with their own history and specific disease course. Therefore, for drugs or treatments, one can generally never guarantee an effect or grant or exclude risks and side effects. Common expectations are summarized below. However, patient-specific deviations are to be expected.
What is Bone Marrow Concentrate - BMC?
Bone Marrow concentrate (BMC) is a source for Mesenchymal Stem Cells (MSCs) and Hematopoietic Stem cells (HSC). It contains many important growth and regenerative factors, in addition to the MSC and HSC in natural composition. The BMC procedure is relatively simple and minimally invasive, therefore it has been a favorite source for stem cell-based therapies in the previous decades. Many clinics rely on BMC as their main stem cell treatment, sometimes with exaggerated claims. However, BMC has demonstrated impressive results for effective treatment of numerous diseases, among them being osteoarthritis. Read more about our BMC Treatment here.
What is Platelet Rich Plasma - PRP?
Platelet Rich Plasma (PRP) is a blood-derived, cellular product with concentrated supply of regenerative growth factors and cytokines, obtained from the patient's own blood. It is very simple to acquire, and it has shown promising results in the treatment of several inflammatory and degenerative diseases. For the treatment of specific diseases, it can be combined with BMC or adMSCs, as it has synergistic additive effects to the treatment. PRP has "special" functions: it serves as a growth medium to maintain stem cells healthy; ensures adequate cellular environment where enough energy is provided to allow the cells to perform their regenerative work.
Is Therapeutic Success Guaranteed?
No therapy can guarantee a 100% success after treatment. However, in the case of experimental therapies such as stem cell therapy, the attending physician must perform a benefit-to-risk analysis for each patient and determine both the benefits and the risks for that particular patient. If the potential benefit outweighs the potential side effects, the doctor may recommend experimental therapy.
References and Literature - Stem Cell-based Therapies and Osteoarthritis
- Zeckser, J., Wolff, M., Tucker, J., & Goodwin, J. (2016). Multipotent Mesenchymal Stem Cell Treatment for Discogenic Low Back Pain and Disc Degeneration, 2016, 4–6.
- Baglio, Serena Rubina, Dirk Michiel Pegtel, and Nicola Baldini. "Mesenchymal stem cell secreted vesicles provide novel opportunities in (stem) cell-free therapy." Frontiers in physiology 3 (2012): 359.
- Richardson, Stephen M., et al. "Mesenchymal stem cells in regenerative medicine: focus on articular cartilage and intervertebral disc regeneration." Methods 99 (2016): 69-80.
- Strassburg, Sandra, et al. "Bi-directional exchange of membrane components occurs during co-culture of mesenchymal stem cells and nucleus pulposus cells." PLoS One 7.3 (2012): e33739.
- K. A. Pettine, M. B. Murphy, R. K. Suzuki, and T. T. Sand, “Percutaneous lumbar intradiscal injection of autologous bone marrowconcentrated cells significantly reduces discogenic pain through 12 months,” Stem Cells, vol. 33, no. 1, pp. 146–156, 2015.
- Mesoblast Reports Positive 24 Month Results in Phase 2 Trial for Chronic Low Back Pain and Initiation of the Phase 3 Program at JP Morgan Healthcare Conference, January 2015, http://www.mesoblast.com. [7] N. Bogduk, C. Aprill, and R. Derby, “Lumbar discogenic pain: state-of-the-art review,” Pain Medicine, vol. 14, no. 6, pp. 813– 836, 2013.
- M. J. DePalma, J. M. Ketchum, and T. R. Saullo, “Etiology of chronic low back pain in patients having undergone lumbar fusion,” Pain Medicine, vol. 12, no. 5, pp. 732–739, 2011.
- J. C. Iatridis, S. B. Nicoll, A. J. Michalek, B. A. Walter, and M. S. Gupta, “Role of biomechanics in intervertebral disc degeneration and regenerative therapies: what needs repairing in the disc and what are promising biomaterials for its repair?” Spine Journal, vol. 13, no. 3, pp. 243–262, 2013.
- N. Bogduk, Practice Guidelines for Spinal Diagnostic and Treatment Procedures, International Spine Intervention Society, San Diego, Calif, USA, 2nd edition, 2013.
- K. M.Malik, S. P.Cohen,D. R.Walega, and H. T. Benzon, “Diagnostic criteria and treatment of discogenic pain: a systematic review of recent clinical literature,” The Spine Journal, vol. 13, no. 11, pp. 1675–1689, 2013.
- U. G. Longo, N. Papapietro, S. Petrillo, E. Franceschetti, N. Maffulli, and V.Denaro, “Mesenchymal stem cell for prevention and management of intervertebral disc degeneration,” Stem Cells International, vol. 2012, Article ID 921053, 7 pages, 2012.
- H. T. J. Gilbert, J. A. Hoyland, and S. M. Richardson, “Stem cell regeneration of degenerated intervertebral discs: current status (Update),” Current Pain and Headache Reports, vol. 17, article 377, 2013.
- H. J. Braun, N. Wilcox-Fogel, H. J. Kim, M. A. Pouliot, A. H. S. Harris, and J. L. Dragoo, “The effect of local anesthetic and corticosteroid combinations on chondrocyte viability,” Knee Surgery, Sports Traumatology, Arthroscopy, vol. 20, no. 9, pp. 1689–1695, 2012.
- J. L. Dragoo, C. M. Danial, H. J. Braun, M. A. Pouliot, and H. J. Kim, “The chondrotoxicity of single-dose corticosteroids,” Knee Surgery, Sports Traumatology, Arthroscopy, vol. 20, no. 9, pp. 1809–1814, 2012.
- B. Peng, X. Pang, Y.Wu, C. Zhao, and X. Song, “A randomized placebo-controlled trial of intradiscal methylene blue injection for the treatment of chronic discogenic lowback pain,” Pain, vol. 149, no. 1, pp. 124–129, 2010.
- S.-H. Kim, S.-H. Ahn, Y.-W. Cho, and D.-G. Lee, “Effect of intradiscal methylene blue injection for the chronic discogenic low back pain: one year prospective follow-up study,” Annals of Rehabilitation Medicine, vol. 36, no. 5, pp. 657–664, 2012.
- W. Yin, K. Pauza, W. J. Olan, J. F. Doerzbacher, and K. J. Thorne, “Intradiscal injection of fibrin sealant for the treatment of symptomatic lumbar internal disc disruption: results of a prospective multicenter pilot study with 24-month follow-up,” Pain Medicine, vol. 15, no. 1, pp. 16–31, 2014.
- S.-Z. Wang, Y.-F. Rui, Q. Tan, and C. Wang, “Enhancing intervertebral disc repair and regeneration through biology: plateletrich plasma as an alternative strategy,” Arthritis Research & Therapy, vol. 15, no. 5, article 220, 2013.
- G. B. Gullung, W. Woodall, M. Tucci, J. James, D. Black, and R. McGuire, “Platelet-rich plasma effects on degenerative disc disease: analysis of histology and imaging in an animal model,” Evidence-Based Spine-Care Journal, vol. 2, no. 4, pp. 13–18, 2011.
- Y.A.Tuakli-Wosornu, A. Terry, K. Boachie-Adjei et al., “Lumbar intradiskal platelet-rich plasma (PRP) injections: a prospective, double-blind, randomized controlled study,” PM&R, 2015.
- L. Manchikanti, F. J. E. Falco, R. M. Benyamin et al., “An update of the systematic assessment of mechanical lumbar disc decompression with nucleoplasty,” Pain Physician, vol. 16, no. 2, pp. SE25–SE54, 2013.
- V. Singh, L. Manchikanti, A. K. Calodney et al., “Percutaneous lumbar laser disc decompression: an update of current evidence,” Pain Physician, vol. 16, no. 2, pp. 229–260, 2013.
- D. H. Jo and H. J. Yang, “The survey of the patient received the epiduroscopic laser neural decompression,” The Korean Journal of Pain, vol. 26, no. 1, pp. 27–31, 2013.
- W. C. H. Jacobs, S. M. Rubinstein, P. C. Willems et al., “The evidence on surgical interventions for low back disorders, an overviewof systematic reviews,” European Spine Journal, vol. 22, no. 9, pp. 1936–1949, 2013.
- W. Shohei, Y. Kuroda, F. Ogura, T. Shigemoto, and M. Dezawa, “Regenerative effects of mesenchymal stem cells: contribution of muse cells, a novel pluripotent stem cell type that resides in mesenchymal cells,” Cells, vol. 1, no. 4, pp. 1045–1060, 2012.
- P. Ghosh, R.Moore, B. Vernon-Roberts et al., “Immunoselected STRO-3+ mesenchymal precursor cells and restoration of the extracellular matrix of degenerate intervertebral discs: laboratory investigation,” Journal of Neurosurgery: Spine, vol. 16, no. 5, pp. 479–488, 2012.
- C. J. Centeno, “Clinical challenges and opportunities of mesenchymal stem cells in musculoskeletal medicine,” PM&R, vol. 6, no. 1, pp. 70–77, 2014.
- A. Cond´e-Green, R. L. Rodriguez, S. Slezak, D. P. Singh, N. H. Goldberg, and J. Mclenithan, “Comparison between stromal vascular cells’ isolation with enzymatic digestion and mechanical processing of aspirated adipose tissue,” Plastic and Reconstructive Surgery, vol. 134, no. 4, p. 54, 2014.
- L.-L. Lu, Y.-J. Liu, S.-G. Yang et al., “Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials,” Haematologica, vol. 91, no. 8, pp. 1017–1026, 2006.
- H. Brisby, N. Papadimitriou, C. Brantsing, P. Bergh, A. Lindahl, and H. Barreto Henriksson, “The presence of local mesenchymal progenitor cells in human degenerated intervertebral discs and possibilities to influence these in vitro: a descriptive study in humans,” Stem Cells and Development, vol. 22, no. 5, pp. 804– 814, 2013.
- M. Tanaka, D. Sakai, A. Hiyama et al., “Effect of cryopreservation on canine and human activated nucleus pulposus cells: a feasibility study for cell therapy of the intervertebral disc,” BioResearch Open Access, vol. 2, no. 4, pp. 273–282, 2013.
- D. Coric, K. Pettine, A. Sumich, and M. O. Boltes, “Prospective study of disc repair with allogeneic chondrocytes,” Journal of Neurosurgery: Spine, vol. 18, no. 1, pp. 85–95, 2013.
- H. J.Meisel, V. Siodla, T. Ganey, Y.Minkus, W. C. Hutton, and O. J. Alasevic, “Clinical experience in cell-based therapeutics: disc chondrocyte transplantation: a treatment for degenerated or damaged intervertebral disc,” Biomolecular Engineering, vol. 24, no. 1, pp. 5–21, 2007.
- T. Miyamoto, T. Muneta, T. Tabuchi et al., “Intradiscal transplantation of synovial mesenchymal stem cells prevents intervertebral disc degeneration through suppression of matrix metalloproteinase-related genes in nucleus pulposus cells in rabbits,”Arthritis Research&Therapy, vol. 12,no. 6, articleR206, 2010.
- S. Strassburg, S. M. Richardson, A. J. Freemont, and J. A. Hoyland, “Co-culture induces mesenchymal stem cell differentiation and modulation of the degenerate human nucleus pulposus cell phenotype,” Regenerative Medicine, vol. 5, no. 5, pp. 701–711, 2010.
- Z. Sun, Z.-H. Liu, X.-H. Zhao et al., “Impact of direct cell cocultures on human adipose-derived stromal cells and nucleus pulposus cells,” Journal of Orthopaedic Research, vol. 31, no. 11, pp. 1804–1813, 2013.
- X. Li, J. P. Lee, G. Balian, and D. G. Anderson, “Modulation of chondrocytic properties of fat-derivedmesenchymal cells in cocultures with nucleus pulposus,”Connective Tissue Research, vol. 46, no. 2, pp. 75–82, 2005.
- C. LeVisage, S.W.Kim,K.Tateno, A.N. Sieber, J.P.Kostuik, and K. W. Leong, “Interaction of human mesenchymal stem cells with disc cells: changes in extracellular matrix biosynthesis,” Spine, vol. 31, no. 18, pp. 2036–2042, 2006.
- A.W. H. Gebraad, S. Miettinen, D.W. Grijpma, and S. P.Haimi, “Human adipose stem cells in chondrogenic differentiation medium without growth factors differentiate towards annulus fibrosus phenotype in vitro,” Macromolecular Symposia, vol. 334, no. 1, pp. 49–56, 2013.
- K.Wuertz, K. Godburn, C. Neidlinger-Wilke, J. Urban, and J. C. Iatridis, “Behavior of mesenchymal stem cells in the chemical microenvironment of the intervertebral disc,” Spine, vol. 33, no. 17, pp. 1843–1849, 2008.
- D. Sakai, J. Mochida, T. Iwashina et al., “Regenerative effects of transplanting mesenchymal stem cells embedded in atelocollagen to the degenerated intervertebral disc,” Biomaterials, vol. 27, no. 3, pp. 335–345, 2006.
- V. Y. L. Leung, D.M. K. Aladin, F. Lv et al., “Mesenchymal stem cells reduce intervertebral disc fibrosis and facilitate repair,” Stem Cells, vol. 32, no. 8, pp. 2164–2177, 2014.
- L. Orozco, R. Soler, C. Morera, M. Alberca, A. S´anchez, and J. Garc´ıa-Sancho, “Intervertebral disc repair by autologous mesenchymal bonemarrowcells: a pilot study,” Transplantation, vol. 92, no. 7, pp. 822–828, 2011.
- Mesoblast Limited, “Positive Spinal Disc Repair Trial Results Using Mesoblast Adult Stem Cells,” 2014, http://www.globenewswire. com/.
- A. Colombini, C. Ceriani, G. Banfi, M. Brayda-Bruno, and M. Moretti, “Fibrin in intervertebral disc tissue engineering,” Tissue Engineering Part B: Reviews, vol. 20, no. 6, pp. 713–721, 2014.
- B. R.Whatley and X.Wen, “Intervertebral disc (IVD): structure, degeneration, repair and regeneration,” Materials Science & Engineering C, vol. 32, no. 2, pp. 61–77, 2012.
- Y. Wang, Z.-B. Han, Y.-P. Song, and Z. C. Han, “Safety of mesenchymal stem cells for clinical application,” Stem Cells International, vol. 2012,Article ID 652034, 4 pages, 2012.
- G.Vadal'a, G. Sowa,M.Hubert, L. G.Gilbertson,V.Denaro, and J. D. Kang, “Mesenchymal stem cells injection in degenerated intervertebral disc: cell leakage may induce osteophyte formation,” Journal of Tissue Engineering and Regenerative Medicine, vol. 6, no. 5, pp. 348–355, 2012.
- W.-H. Chen, H.-Y. Liu, W.-C. Lo et al., “Intervertebral disc regeneration in an ex vivo culture system using mesenchymal stem cells and platelet-rich plasma,” Biomaterials, vol. 30, no. 29, pp. 5523–5533, 2009.
- Y. Zhu, M. Yuan, H. Y. Meng et al., “Basic science and clinical application of platelet-rich plasma forcartilage defects and osteoarthritis: a review,” Osteoarthritis and Cartilage, vol. 21, no. 11, pp. 1627–1637, 2013.
- C. S. Lee,O. A. Burnsed,V. Raghuram, J.Kalisvaart, B.D. Boyan, and Z. Schwartz, “Adipose stem cells can secrete angiogenic factors that inhibit hyaline cartilage regeneration,” Stem Cell Research &Therapy, vol. 3, no. 4, article 35, 2012.
- T. E. Foster, B. L. Puskas, B. R. Mandelbaum, M. B. Gerhardt, and S. A. Rodeo, “Platelet-rich plasma: from basic science to clinical applications,” The American Journal of Sports Medicine, vol. 37, no. 11, pp. 2259–2272, 2009.
- A. S.Wasterlain, H. J. Braun, A. H. S. Harris, H.-J. Kim, and J. L. Dragoo, “The systemic effects of platelet-rich plasma injection,” The American Journal of Sports Medicine, vol. 41, no. 1, pp. 186– 193, 2013.
Further References for MSC, BMC, Stemcell Secretome and EVs
- Georg Hansmann, Philippe Chouvarine, Franziska Diekmann, Martin Giera, Markus Ralser, Michael Mülleder, Constantin von Kaisenberg, Harald Bertram, Ekaterina Legchenko & Ralf Hass "Human umbilical cord mesenchymal stem cell-derived treatment of severe pulmonary arterial hypertension". Nature Cardiovascular Research volume 1, pages568–576 (2022).
- Murphy JM, Fink DJ, Hunziker EB, et al. Stem cell therapy in a caprine model of osteoarthritis . Arthritis Rheum. 2003;48:3464–74.
- Lee KB, Hui JH, Song IC, Ardany L, et al. Injectable mesenchymal stem cell therapy for large cartilage defects—a porcine model. Stem Cell. 2007;25:2964–71.
- Saw KY, Hussin P, Loke SC, et al. Articular cartilage regeneration with autologous marrow aspirate and hyaluronic acid: an experimental study in a goat model. Arthroscopy . 2009;25(12):1391–400.
- Black L, Gaynor J, Adams C, et al. Effect of intra-articular injection of autologous adipose-derived mesenchymal stem and regenerative cells on clinical signs of chronic osteoarthritis of the elbow joint in dogs. Vet Ther. 2008;9:192-200.
- Centeno C, Busse D, Kisiday J, et al. Increased knee cartilage volume in degenerative joint disease using percutaneously implanted, autologous mesenchymal stem cells. Pain Physician. 2008;11(3):343–53.
- Centeno C, Kisiday J, Freeman M, et al. Partial regeneration of the human hip via autologous bone marrow nucleated cell transfer: a case study. Pain Physician. 2006;9:253–6.
- Centeno C, Schultz J, Cheever M. Safety and complications reporting on the re-implantation of culture-expanded mesenchymal stem cells using autologous platelet lysate technique. Curr Stem Cell. 2011;5(1):81–93.
- Pak J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose derived stem cells: a case series. J Med Case Rep. 2001;5:296.
- Kuroda R, Ishida K, et al. Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage. 2007;15:226–31.
- Emadedin M, Aghdami N, Taghiyar L, et al. Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch Iran Med. 2012;15(7):422–8.
- Saw KY et al. Articular cartilage regeneration with autologous peripheral blood stem cells versus hyaluronic acid: a randomized controlled trial. Arthroscopy. 2013;29(4):684–94.
- Vangsness CT, Farr J, Boyd J, et al. Adult human mesenchymal stem cells delivered via intra-articular injection to the knee following partial medial meniscectomy. J Bone Joint Surg. 2014;96(2):90–8.
- Freitag, Julien, et al. Mesenchymal stem cell therapy in the treatment of osteoarthritis: reparative pathways, safety and efficacy–a review. BMC musculoskeletal disorders 17.1 (2016): 230.
- Maumus, Marie, Christian Jorgensen, and Danièle Noël. " Mesenchymal stem cells in regenerative medicine applied to rheumatic diseases: role of secretome and exosomes. " Biochimie 95.12 (2013): 2229-2234.
- Dostert, Gabriel, et al. " How do mesenchymal stem cells influence or are influenced by microenvironment through extracellular vesicles communication?. " Frontiers in Cell and Developmental Biology 5 (2017).
- Chaparro, Orlando, and Itali Linero. " Regenerative Medicine: A New Paradigm in Bone Regeneration. " (2016).
- Toh, Wei Seong, et al. " MSC exosome as a cell-free MSC therapy for cartilage regeneration: Implications for osteoarthritis treatment. " Seminars in Cell & Developmental Biology. Academic Press, 2016.
- Chaparro, Orlando, and Itali Linero. " Regenerative Medicine: A New Paradigm in Bone Regeneration. " (2016).
- S. Koelling, J. Kruegel, M. Irmer, J.R. Path, B. Sadowski, X. Miro, et al., Migratory chondrogenic progenitor cells from repair tissue during the later stages of human osteoarthritis , Cell Stem Cell 4 (2009) 324–335.
- B.A. Jones, M. Pei, Synovium-Derived stem cells: a tissue-Specific stem cell for cartilage engineering and regeneration , Tissue Eng. B: Rev. 18 (2012) 301–311.
- W. Ando, J.J. Kutcher, R. Krawetz, A. Sen, N. Nakamura, C.B. Frank, et al., Clonal analysis of synovial fluid stem cells to characterize and identify stable mesenchymal stromal cell/mesenchymal progenitor cell phenotypes in a porcine model: a cell source with enhanced commitment to the chondrogenic lineage, Cytotherapy 16 (2014) 776–788.
- K.B.L. Lee, J.H.P. Hui, I.C. Song, L. Ardany, E.H. Lee, Injectable mesenchymal stem cell therapy for large cartilage defects—a porcine model, Stem Cells 25 (2007) 2964–2971.
- W.-L. Fu, C.-Y. Zhou, J.-K. Yu, A new source of mesenchymal stem cells for articular cartilage repair: mSCs derived from mobilized peripheral blood share similar biological characteristics in vitro and chondrogenesis in vivo as MSCs from bone marrow in a rabbit model , Am. J. Sports Med. 42 (2014) 592–601.
- X. Xie, Y. Wang, C. Zhao, S. Guo, S. Liu, W. Jia, et al., Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration , Biomaterials 33 (2012) 7008–7018.
- E.-R. Chiang, H.-L. Ma, J.-P. Wang, C.-L. Liu, T.-H. Chen, S.-C. Hung, Allogeneic mesenchymal stem cells in combination with hyaluronic acid for the treatment of osteoarthritis in rabbits , PLoS One 11 (2016) e0149835.
- H. Nejadnik, J.H. Hui, E.P. Feng Choong, B.-C. Tai, E.H. Lee, Autologous bone marrow–derived mesenchymal stem cells versus autologous chondrocyte implantation: an observational cohort study , Am. J. Sports Med. 38 (2010) 1110–1116.
- I. Sekiya, T. Muneta, M. Horie, H. Koga, Arthroscopic transplantation of synovial stem cells improves clinical outcomes in knees with cartilage defects , Clin. Orthop. Rel. Res. 473 (2015) 2316–2326.
- Y.S. Kim, Y.J. Choi, Y.G. Koh, Mesenchymal stem cell implantation in knee osteoarthritis: an assessment of the factors influencing clinical outcomes , Am. J. Sports Med. 43 (2015) 2293–2301.
- W.-L. Fu, Y.-F. Ao, X.-Y. Ke, Z.-Z. Zheng, X. Gong, D. Jiang, et al., Repair of large full-thickness cartilage defect by activating endogenous peripheral blood stem cells and autologous periosteum flap transplantation combined with patellofemoral realignment , Knee 21 (2014) 609–612.
- Y.-G. Koh, O.-R. Kwon, Y.-S. Kim, Y.-J. Choi, D.-H. Tak, Adipose-derived mesenchymal stem cells with microfracture versus microfracture alone: 2-year follow-up of a prospective randomized trial , Arthrosc. J. Arthrosc. Relat. Surg. 32 (2016) 97–109.
- T.S. de Windt, L.A. Vonk, I.C.M. Slaper-Cortenbach, M.P.H. van den Broek, R. Nizak, M.H.P. van Rijen, et al., Allogeneic mesenchymal stem cells stimulate cartilage regeneration and are safe for single-Stage cartilage repair in humans upon mixture with recycled autologous chondrons , Stem Cells (2016) (n/a-n/a).
- L. da Silva Meirelles, A.M. Fontes, D.T. Covas, A.I. Caplan, Mechanisms involved in the therapeutic properties of mesenchymal stem cells , Cytokine Growth Factor Rev. 20 (2009) 419–427.
- W.S. Toh, C.B. Foldager, M. Pei, J.H.P. Hui, Advances in mesenchymal stem cell-based strategies for cartilage repair and regeneration , Stem Cell Rev. Rep. 10 (2014) 686–696.
- R.C. Lai, F. Arslan, M.M. Lee, N.S.K. Sze, A. Choo, T.S. Chen, et al., Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury , Stem Cell Res. 4 (2010) 214–222.
- S. Zhang, W.C. Chu, R.C. Lai, S.K. Lim, J.H.P. Hui, W.S. Toh, Exosomes derived from human embryonic mesenchymal stem cells promote osteochondral regeneration, Osteoarthr . Cartil. 24 (2016) 2135–2140.
- S. Zhang, W. Chu, R. Lai, J. Hui, E. Lee, S. Lim, et al., 21 – human mesenchymal stem cell-derived exosomes promote orderly cartilage regeneration in an immunocompetent rat osteochondral defect model , Cytotherapy 18 (2016) S13.
- C.T. Lim, X. Ren, M.H. Afizah, S. Tarigan-Panjaitan, Z. Yang, Y. Wu, et al., Repair of osteochondral defects with rehydrated freeze-dried oligo[poly(ethylene glycol) fumarate] hydrogels seeded with bone marrow mesenchymal stem cells in a porcine model
- A. Gobbi, G. Karnatzikos, S.R. Sankineani, One-step surgery with multipotent stem cells for the treatment of large full-thickness chondral defects of the knee , Am. J. Sports Med. 42 (2014) 648–657.
- A. Gobbi, C. Scotti, G. Karnatzikos, A. Mudhigere, M. Castro, G.M. Peretti, One-step surgery with multipotent stem cells and Hyaluronan-based scaffold for the treatment of full-thickness chondral defects of the knee in patients older than 45 years , Knee Surg. Sports Traumatol. Arthrosc. (2016) 1–8.
- A. Gobbi, G. Karnatzikos, C. Scotti, V. Mahajan, L. Mazzucco, B. Grigolo, One-step cartilage repair with bone marrow aspirate concentrated cells and collagen matrix in full-thickness knee cartilage lesions: results at 2-Year follow-up , Cartilage 2 (2011) 286–299.
- K.L. Wong, K.B.L. Lee, B.C. Tai, P. Law, E.H. Lee, J.H.P. Hui, Injectable cultured bone marrow-derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years’ follow-up , Arthrosc. J. Arthrosc. Relat. Surg. 29 (2013) 2020–2028.
- J.M. Hare, J.E. Fishman, G. Gerstenblith, et al., Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the poseidon randomized trial, JAMA 308 (2012) 2369–2379.
- L. Wu, J.C.H. Leijten, N. Georgi, J.N. Post, C.A. van Blitterswijk, M. Karperien, Trophic effects of mesenchymal stem cells increase chondrocyte proliferation and matrix formation , Tissue Eng. A 17 (2011) 1425–1436.
- L. Wu, H.-J. Prins, M.N. Helder, C.A. van Blitterswijk, M. Karperien, Trophic effects of mesenchymal stem cells in chondrocyte Co-Cultures are independent of culture conditions and cell sources , Tissue Eng. A 18 (2012) 1542–1551.
- S.K. Sze, D.P.V. de Kleijn, R.C. Lai, E. Khia Way Tan, H. Zhao, K.S. Yeo, et al., Elucidating the secretion proteome of human embryonic stem cell-derived mesenchymal stem cells , Mol. Cell. Proteomics 6 (2007) 1680–1689.
- M.B. Murphy, K. Moncivais, A.I. Caplan, Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine , Exp. Mol. Med. 45 (2013) e54.
- M.J. Lee, J. Kim, M.Y. Kim, Y.-S. Bae, S.H. Ryu, T.G. Lee, et al., Proteomic analysis of tumor necrosis factor--induced secretome of human adipose tissue-derived mesenchymal stem cells , J. Proteome Res. 9 (2010) 1754–1762.
- S. Bruno, C. Grange, M.C. Deregibus, R.A. Calogero, S. Saviozzi, F. Collino, et al., Mesenchymal stem cell-derived microvesicles protect against acute tubular injury, J. Am. Soc. Nephrol. 20 (2009) 1053–1067.
- M. Yá˜nez-Mó, P.R.-M. Siljander, Z. Andreu, A.B. Zavec, F.E. Borràs, E.I. Buzas, et al. Biological properties of extracellular vesicles and their physiological functions (2015).
- C. Lawson, J.M. Vicencio, D.M. Yellon, S.M. Davidson, Microvesicles and exosomes: new players in metabolic and cardiovascular disease , J. Endocrinol. 228 (2016) R57–R71.
- A.G. Thompson, E. Gray, S.M. Heman-Ackah, I. Mager, K. Talbot, S.E. Andaloussi, et al., Extracellular vesicles in neurodegenerative diseas—pathogenesis to biomarkers, Nat. Rev. Neurol. 12 (2016) 346–357.
- I.E.M. Bank, L. Timmers, C.M. Gijsberts, Y.-N. Zhang, A. Mosterd, J.-W. Wang, et al., The diagnostic and prognostic potential of plasma extracellular vesicles for cardiovascular disease , Expert Rev. Mol. Diagn. 15 (2015) 1577–1588.
- T. Kato, S. Miyaki, H. Ishitobi, Y. Nakamura, T. Nakasa, M.K. Lotz, et al., Exosomes from IL-1 stimulated synovial fibroblasts induce osteoarthritic changes in articular chondrocytes , Arthritis. Res. Ther. 16 (2014) 1–11.
- R.W.Y. Yeo, S.K. Lim, Exosomes and their therapeutic applications, in: C. Gunther, A. Hauser, R. Huss (Eds.), Advances in Pharmaceutical Cell TherapyPrinciples of Cell-Based Biopharmaceuticals, World Scientific, Singapore, 2015, pp. 477–491.
- X. Qi, J. Zhang, H. Yuan, Z. Xu, Q. Li, X. Niu, et al., Exosomes secreted by human-Induced pluripotent stem cell-derived mesenchymal stem cells repair critical-sized bone defects through enhanced angiogenesis and osteogenesis in osteoporotic rats , Int. J. Biol. Sci. 12 (2016) 836–849.
- R.C. Lai, F. Arslan, S.S. Tan, B. Tan, A. Choo, M.M. Lee, et al., Derivation and characterization of human fetal MSCs: an alternative cell source for large-scale production of cardioprotective microparticles , J. Mol. Cell. Cardiol. 48 (2010) 1215–1224.
- Y. Zhou, H. Xu, W. Xu, B. Wang, H. Wu, Y. Tao, et al., Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro , Stem Cell Res. Ther. 4 (2013) 1–13.
- Y. Qin, L. Wang, Z. Gao, G. Chen, C. Zhang, Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo , Sci. Rep. 6 (2016) 21961.
- M. Nakano, K. Nagaishi, N. Konari, Y. Saito, T. Chikenji, Y. Mizue, et al., Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by exosome transfer into damaged neurons and astrocytes , Sci. Rep. 6 (2016) 24805.
- K. Nagaishi, Y. Mizue, T. Chikenji, M. Otani, M. Nakano, N. Konari, et al., Mesenchymal stem cell therapy ameliorates diabetic nephropathy via the paracrine effect of renal trophic factors including exosomes , Sci. Rep. 6 (2016) 34842.
- S.R. Baglio, K. Rooijers, D. Koppers-Lalic, F.J. Verweij, M. Pérez Lanzón, N. Zini, et al., Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species , Stem Cell Res. Ther. 6 (2015) 1–20.
- T. Chen, R. Yeo, F. Arslan, Y. Yin, S. Tan, Efficiency of exosome production correlates inversely with the developmental maturity of MSC donor, J. Stem Cell Res. Ther. 3 (2013) 2.
- R.C. Lai, S.S. Tan, B.J. Teh, S.K. Sze, F. Arslan, D.P. de Kleijn, et al., Proteolytic potential of the MSC exosome proteome: implications for an exosome-mediated delivery of therapeutic proteasome , Int. J. Proteomics 2012 (2012) 971907.
- T.S. Chen, R.C. Lai, M.M. Lee, A.B.H. Choo, C.N. Lee, S.K. Lim, Mesenchymal stem cell secretes microparticles enriched in pre-microRNAs , Nucleic Acids Res. 38 (2010) 215–224.
- R.W. Yeo, R.C. Lai, K.H. Tan, S.K. Lim, Exosome: a novel and safer therapeutic refinement of mesenchymal stem cell, J. Circ. Biomark. 1 (2013) 7.
- R.C. Lai, R.W. Yeo, S.K. Lim, Mesenchymal stem cell exosomes, Semin. Cell Dev. Biol. 40 (2015) 82–88.
- B. Zhang, R.W. Yeo, K.H. Tan, S.K. Lim, Focus on extracellular vesicles: therapeutic potential of stem cell-derived extracellular vesicles , Int. J. Mol. Sci. 17 (2016) 174.
- Hu G-w, Q. Li, X. Niu, B. Hu, J. Liu, Zhou S-m, et al., Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice , Stem Cell Res. Ther. 6 (2015) 1–15.
- J. Zhang, J. Guan, X. Niu, G. Hu, S. Guo, Q. Li, et al., Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis , J. Transl. Med. 13 (2015) 1–14.
- B. Zhang, M. Wang, A. Gong, X. Zhang, X. Wu, Y. Zhu, et al., HucMSC-exosome mediated-Wnt4 signaling is required for cutaneous wound healing, Stem Cells 33 (2015) 2158–2168.
- B. Zhang, Y. Yin, R.C. Lai, S.S. Tan, A.B.H. Choo, S.K. Lim, Mesenchymal stem cells secrete immunologically active exosomes , Stem Cells Dev. 23 (2013) 1233–1244.
- C.Y. Tan, R.C. Lai, W. Wong, Y.Y. Dan, S.-K. Lim, H.K. Ho, Mesenchymal stem cell-derived exosomes promote hepatic regeneration in drug-induced liver injury models , Stem Cell Res. Ther. 5 (2014) 1–14.
- C. Lee, S.A. Mitsialis, M. Aslam, S.H. Vitali, E. Vergadi, G. Konstantinou, et al., Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia-induced pulmonary hypertension , Circulation 126 (2012) 2601–2611.
- B. Yu, H. Shao, C. Su, Y. Jiang, X. Chen, L. Bai, et al., Exosomes derived from MSCs ameliorate retinal laser injury partially by inhibition of MCP-1 , Sci. Rep. 6 (2016) 34562.
- Jo CH, Lee YG, Shin WH, et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof of concept clinical trial. Stem Cells. 2014;32(5):1254–66.
- Vega, Aurelio, et al. Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation. 2015;99(8):1681–90.
- Davatchi F, Sadeghi-Abdollahi B, Mohyeddin M, et al. Mesenchymal stem cell therapy for knee osteoarthritis. Preliminary report of four patients. Int J Rheum Dis. 2011;14(2):211–5
- Hernigou P, Flouzat Lachaniette CH, Delambre J, et al. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case- controlled study. Int Orthop. 2014;38(9):1811–1818
- Galli D, Vitale M, Vaccarezza M. Bone marrow-derived mesenchymal cell differentiation toward myogenic lineages: facts and perspectives. Biomed Res Int. 2014;2014:6.
- Beitzel K, Solovyova O, Cote MP, et al. The future role of mesenchymal Stem cells in The management of shoulder disorders . Arthroscopy. 2013;29(10):1702–1711.
- Isaac C, Gharaibeh B, Witt M, Wright VJ, Huard J. Biologic approaches to enhance rotator cuff healing after injury. J Shoulder Elbow Surg. 2012;21(2):181–190.
- Malda, Jos, et al. " Extracellular vesicles [mdash] new tool for joint repair and regeneration. " Nature Reviews Rheumatology (2016).
Further References about PRP
- Rubio-Azpeitia E, Andia I. Partnership between platelet-rich plasma and mesenchymal stem cells: in vitro experience. Muscles Ligaments Tendons J. 2014;4(1):52–62.
Extras
- Xu, Ming, et al. " Transplanted senescent cells induce an osteoarthritis-like condition in mice. " The Journals of Gerontology Series A: Biological Sciences and Medical Sciences (2016): glw154.
- McCulloch, Kendal, Gary J. Litherland, and Taranjit Singh Rai. " Cellular senescence in osteoarthritis pathology ." Aging Cell (2017).
Contraindications
Our stem cell treatments are experimental, but we only treat patients for whom we believe the risk/benefit ratio indicates treatment based on the state of the art, i.e., medical, scientific evidence.
Please understand that we therefore do not treat patients for whom the following points apply:
- Active cancer in the last two years
- Not yet of legal age
- Existing pregnancy or lactation period
- Unable to breathe on own, ventilator
- Difficulty breathing in supine position
- Dysphagia (extreme difficulty swallowing)
- Psychiatric disorder
- Active infectious disease (Hepatitis A, B, C, HIV, Syphilis, or other)